| Product Name | HSP90 Alpha ELISA kit |
| Description |
Colorimetric detection of HSP90 Alpha |
| Species Reactivity | Avian (Zebra finch), Goat, Human |
| Platform | Microplate |
| Sample Types | Cell lysates, Serum, Tissue, Whole Blood |
| Detection Method | Colorimetric Assay |
| Assay Type | Sandwich ELISA (Enzyme-linked Immunosorbent Assay) |
| Utility | ELISA kit used to quantitate HSP90 alpha concentration in samples. |
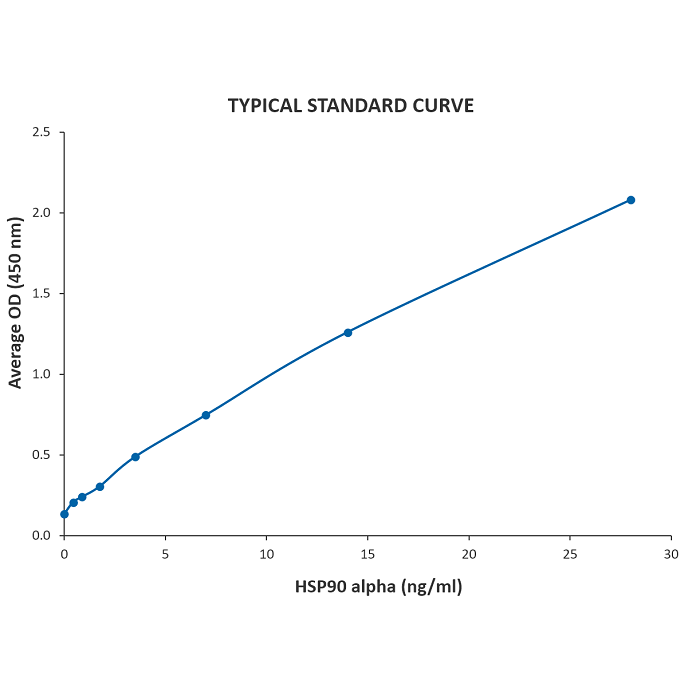
| Sensitivity | 0.117 ng/ml |
| Assay Range | 0.44 - 28 ng/ml |
| Incubation Time | 30 minutes |
| Number of Samples | 40 samples in duplicate |
| Other Resources | Kit Booklet Lot No. SH531948 , Kit Booklet Lot No. SH187452 , Kit Booklet Lot No. SH388280 , Kit Booklet Lot No. KH387128 , MSDS |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | 4ºC and -20ºC | ||||||||||||||||||||||||||||||||||||
| Shipping Temperature | Blue Ice | ||||||||||||||||||||||||||||||||||||
| Product Type | ELISA Kits | ||||||||||||||||||||||||||||||||||||
| Assay Overview | 1. Prepare Standard and samples in Standard and Sample Diluent. 2. Add 100 µL of Standard or sample to appropriate wells. 3. Cover plate with Plate Sealer and incubate at 37°C for 1 hour. 4. Wash plate four times with 1X Wash Buffer. 5. Add 100 µL of Biotinylated Antibody Working Solution to each well. 6. Cover plate with Plate Sealer and incubate at room temperature, 20-25 °C for 1 hour. 7. Wash plate four times with 1X Wash Buffer. 8. Add 100 µL of Streptavidin-HRP Working Solution to each well. 9. Cover plate with Plate Sealer and incubate at room temperature for 30 minutes. 10. Wash plate four times with 1X Wash Buffer. 11. Add 100 µL of TMB Substrate to each well. 12. Develop the plate in the dark at room temperature for 30 minutes. 13. Stop reaction by adding 100 µL of Stop Solution to each well. 14. Measure absorbance on a plate reader at 450 nm. | ||||||||||||||||||||||||||||||||||||
| Kit Overview |
|
||||||||||||||||||||||||||||||||||||
| Cite This Product | HSP90 alpha ELISA Kit (StressMarq Biosciences Inc., Victoria BC CANADA, Catalog # SKT-107) |
Biological Description
| Alternative Names | HSP86 ELISA Kit, HSP89A ELISA Kit, HSP90A ELISA Kit, HSP90AA1 ELISA Kit, HSPC1 ELISA Kit, HSPCA ELISA Kit, HsoCAL3 ELISA Kit, HSP90alpha ELISA Kit |
| Research Areas | Cancer, Cell Signaling, Chaperone Proteins, Heat Shock, Protein Trafficking, Tumor Biomarkers |
| Scientific Background | HSP90 is a highly conserved and essential stress protein that is expressed in all eukaryotic cells. From a functional perspective, HSP90 participates in the folding, assembly, maturation, and stabilization of specific proteins as an integral component of a chaperone complex (1-4). Despite its label of being a heat-shock protein, HSP90 is one of the most highly expressed proteins in unstressed cells (1–2% of cytosolic protein). It carries out a number of housekeeping functions – including controlling the activity, turnover, and trafficking of a variety of proteins. Most of the HSP90- regulated proteins that have been discovered to date are involved in cell signaling (5-6). The number of proteins now known to interact with HSP90 is about 100. Target proteins include the kinases v-Src, Wee1, and c-Raf, transcriptional regulators such as p53 and steroid receptors, and the polymerases of the hepatitis B virus and telomerase(3). When bound to ATP, HSP90 interacts with co-chaperones Cdc37, p23, and an assortment of immunophilin-like proteins, forming a complex that stabilizes and protects target proteins from proteasomal degradation. In most cases, HSP90-interacting proteins have been shown to co-precipitate with HSP90 when carrying out immune-oadsorption studies, and to exist in cytosolic heterocomplexes with it. In a number of cases, variations in HSP90 expression or HSP90 mutation has been shown to degrade signaling function via the protein or to impair a specific function of the protein (such as steroid binding, kinase activity) in vivo. Ansamycin antibiotics, such as geldanamycin and radicicol, inhibit HSP90 function (7). Looking for more information on HSP90? Visit our new HSP90 Scientific Resource Guide at http://www.HSP90.ca. |
| References |
1. Arlander S.J.H., et al. (2003) J Biol Chem. 278: 52572-52577. 2. Pearl H., et al. (2001) Adv Protein Chem. 59: 157-186. 3. Neckers L, et al. (2002) Trends Mol Med. 8:S55-S61. 4. Pratt W., Toft D. (2003) Exp Biol Med. 228:111-133. 5. Pratt W., Toft D. (1997) Endocr Rev. 18: 306-360. 6. Pratt W.B. (1998) Proc Soc Exptl Biol Med. 217: 420-434. 7. Whitesell L., et al. (1994) Proc Natl Acad Sci USA. 91: 8324- 8328. |

StressMarq Biosciences :
Based on validation through cited publications.