| Product Name | Hydrogen Peroxide Detection Kit (Discontinued) |
| Description |
Quantitative colorimetric measurement of H2O2 |
| Species Reactivity | Species Independent |
| Platform | Microplate |
| Sample Types | Buffer, Tissue Culture Media, Urine |
| Detection Method | Colorimetric Assay |
| Assay Type | Direct Quantitative Assay |
| Utility | Colorimetric assay used to quantitatively measure H2O2 in a variety of samples. |
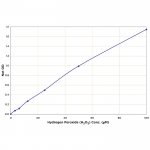
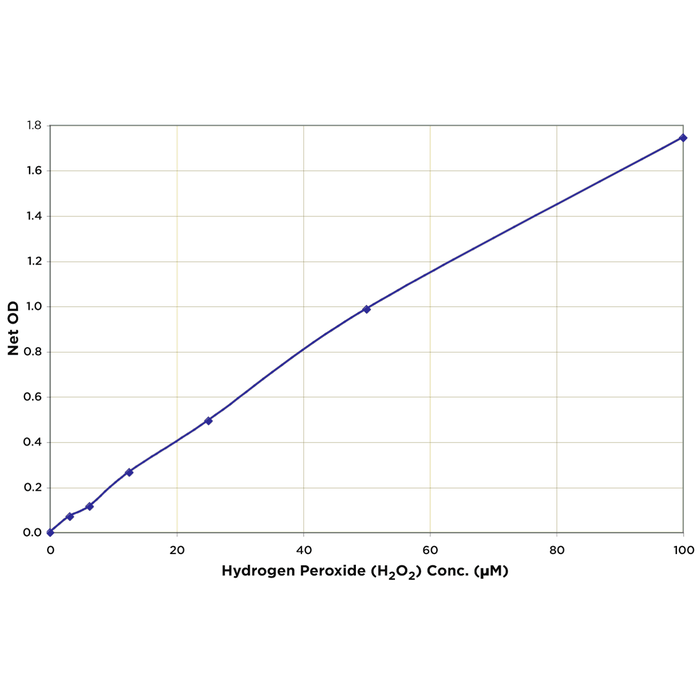
| Sensitivity | 1.83 µM |
| Assay Range | 3.125 - 100 µM |
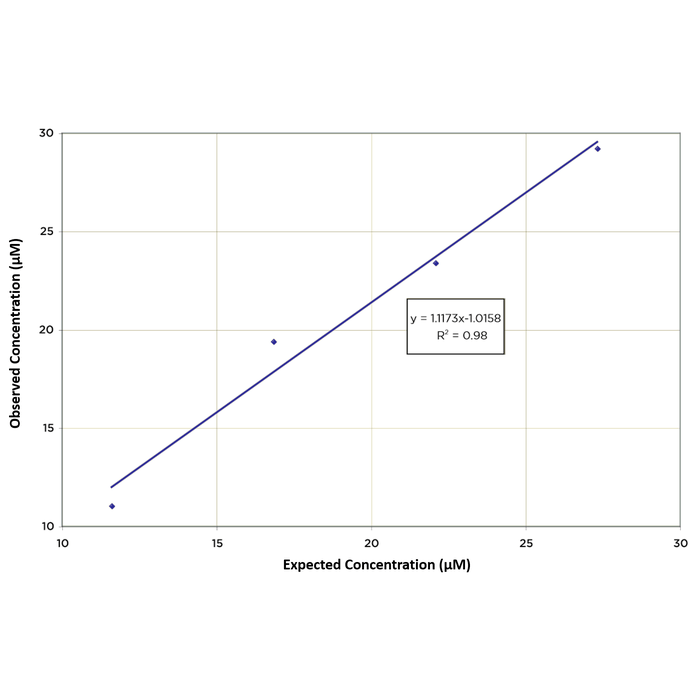
| Precision | Intra Assay Precision: Three buffer samples were run in replicates of 20 in an assay. The mean and precision of the calculated concentrations were: Sample 1- 82.2 µM, 2.1% CV Sample 2- 53.1 µM, 2.4% CV Sample 3- 19.4 µM, 5.9% CV Inter Assay Precision: Three buffer samples were run in duplicate in twelve assays run over multiple days by three operators. The mean and precision of the calculated concentrations were: Sample 1- 79.9 µM, 3.7% CV Sample 2- 49.5 µM, 4.5% CV Sample 3- 18.4 µM, 4.3% CV |
| Incubation Time | 15 Minutes |
| Number of Samples | 89 samples in duplicate |
| Other Resources | Kit Booklet , MSDS |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | 4ºC | ||||||||||||||||||
| Shipping Temperature | Blue Ice | ||||||||||||||||||
| Product Type | Detection Kits | ||||||||||||||||||
| Assay Overview | The Hydrogen Peroxide Detection Kit is designed to quantitatively measure H2O2 in a variety of samples. A hydrogen peroxide standard is provided to generate a standard curve for the assay and all samples should be read off the standard curve. Samples are mixed with the Colorimetric Substrate and the reaction initiated by addition of horseradish peroxidase. The reaction is incubated at room temperature for 15 minutes. The HRP reacts with the substrate in the presence of hydrogen peroxide to convert the colorless substrate into a colored product. The pink product is read at 560 nm. Increasing levels of H2O2 cause a linear increase in color. | ||||||||||||||||||
| Kit Overview |
|
||||||||||||||||||
| Cite This Product | Hydrogen Peroxide Detection Kit (StressMarq Biosciences Inc., Victoria BC CANADA, Catalog # SKT-216) |
Biological Description
| Alternative Names | Dioxidane Detection Kit, Oxidanyl Detection Kit |
| Research Areas | Cancer, Cell Signaling, Oxidation, Oxidative Stress, Post-translational Modifications |
| Scientific Background | In biological systems incomplete reduction of O2 during respiration produces superoxide anion (O2-·), which is spontaneously or enzymatically dismutated by superoxide dismutase to H2O2. Many cells produce low levels of O2-· and H2O2 in response to a variety of extracellular stimuli, such as cytokines (TGF-ß1, TNF-a, and various interleukins), peptide growth factors (PDGF; EGF, VEGF, bFGF, and insulin), the agonists of heterotrimeric G protein–coupled receptors (GPCR) such as angiotensin II, thrombin, lysophosphatidic acid, sphingosine 1-phosphate, histamine, and bradykinin, and by shear stress (1). The addition of exogenous H2O2 or the intracellular production in response to receptor stimulation affects the function of various proteins, including protein kinases, protein phosphatases, transcription factors, phospholipases, ion channels, and G proteins. In 1894, Fenton (2) described the oxidation of tartaric acid by Fe2+ and H2O2. H2O2 and O2 may participate in the production of singlet oxygen and peroxynitrite and the generation of these species may be concurrent with reactions involving iron, and under some circumstances they might be important contributors to H2O2 toxicity (3,4). A substantial portion of H2O2 lethality involves DNA damage by oxidants generated from iron-mediated Fenton reactions (5,6). Damage by Fenton oxidants occurs at the DNA bases or at the sugar residues. Sugar damage is initiated by hydrogen abstraction from one of the deoxyribose carbons, and the predominant consequence is eventual strand breakage and base release (7,8). |
| References |
1. Rhee SG, Bae YS, Lee SR, Kwon J. (2000) Science’s stke. Available at: http://stke.sciencemag.org/cgi/content/abstract/sigtrans;2000/53/pe1 2. Fenton, HJH. J. Chem. Soc. (Lond.) 1894, 65:899–910. 3. Sies, H. Mutat. Res., 1993, 299:183–191. 4. Squadrito, GL., and Pryor, WA. (1995) Chem. Biol. Interact. 96:203–206. 5. Imlay, JA., and Linn, S. (1988) Science 240:1302–1309. 6. Mello-Filho, AC., Meneghini, R. (1991) Mutat. Res., 251:109–113. 7. von Sonntag, C. (1987) pp. 238–249, Taylor and Francis, New York. 8. Henle, ES., Roots, R., Holley, WR., and Chatterjee, A. (1995) Radiat. Res. 143:144–150. |


Reviews
There are no reviews yet.