| Product Name | SOD1 Pre-formed Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
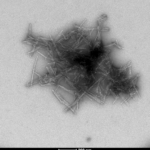
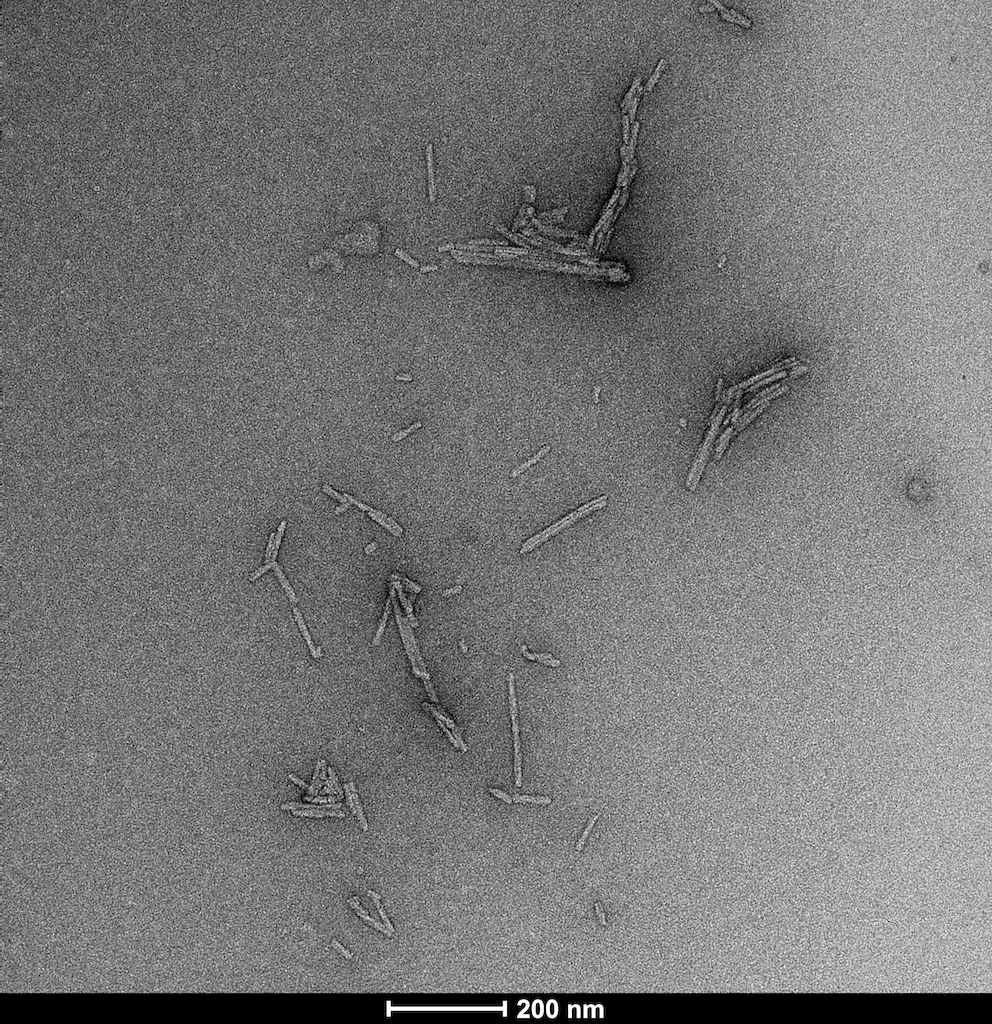
Human Recombinant Superoxide Dismutase 1 (SOD1) PFFs |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS-PAGE, In vivo assay, In vitro assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | Lot/batch specific. See included datasheet. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | Full Length | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | 15.936 kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | PB pH 7.4, 5 mM EDTA, 50 mM DTT |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant SOD1 Pre-formed Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-470) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. |
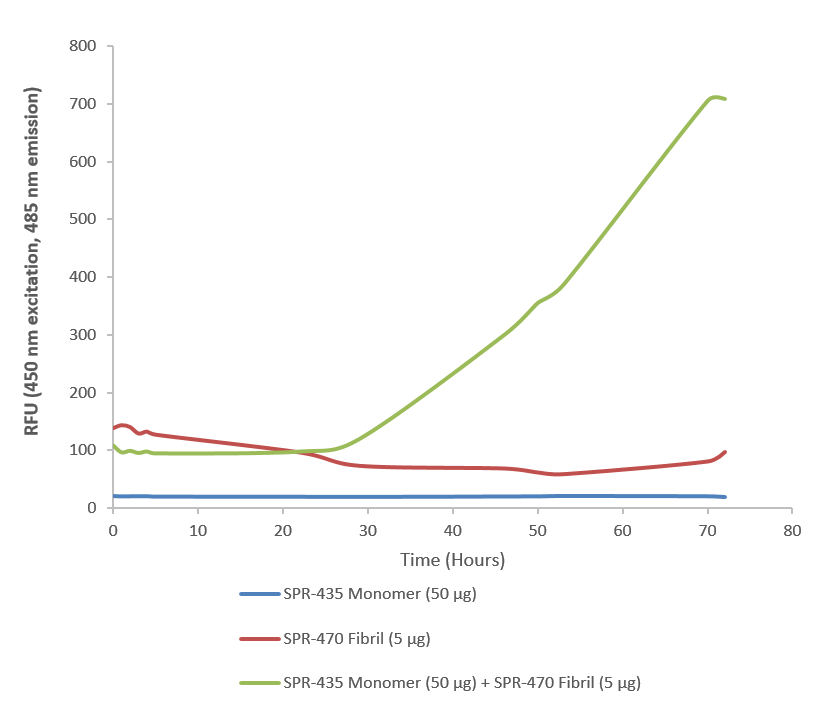
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-435. |
Biological Description
| Alternative Names | Superoxide dismutase 1, SOD1, ALS1, IPOA, Cu/Zn superoxide dismutase, Superoxide dismutase [Cu-Zn], hSOD1, SODC, Cytosolic superoxide dismutase, Erythrocuprein, Hemocuprein, Cytocuprein, Indophenoloxidase A, SOD1 PFFs |
| Research Areas | ALS Disease, Cancer, Cell Signaling, Chaperone Proteins, Neurodegeneration, Neuroscience, Oxidative Stress, Protein Trafficking |
| Cellular Localization | Cytoplasm, Mitochondrion, Nucleus |
| Accession Number | NP_000445.1 |
| Gene ID | 6647 |
| Swiss Prot | P00441 |
| Scientific Background |
Superoxide dismutase 1 (SOD1) is a copper/zinc-binding enzyme essential for cellular antioxidant defense, catalyzing the conversion of superoxide radicals into oxygen and hydrogen peroxide. Mutations or misfolding of SOD1 are strongly linked to amyotrophic lateral sclerosis (ALS), a fatal neurodegenerative disorder characterized by progressive motor neuron degeneration. In ALS, aberrant SOD1 conformations aggregate into toxic species, disrupting protein homeostasis and driving neuronal death. SOD1 Pre-formed Fibrils (PFFs) have emerged as powerful tools for modeling this pathogenic process. These synthetic fibrillar aggregates mimic the misfolded state of SOD1 observed in ALS and can seed the aggregation of endogenous SOD1 in vitro and in vivo. By initiating a cascade of protein misfolding, PFFs replicate key features of ALS pathology, including prion-like propagation and inclusion formation. This makes them invaluable for studying molecular mechanisms of aggregation, oxidative stress, and neurotoxicity. Researchers use SOD1 PFFs to develop robust cellular and animal models, enabling high-throughput screening of therapeutic candidates aimed at preventing or reversing aggregation. Beyond ALS, these models provide insights into shared pathways of protein misfolding implicated in other neurodegenerative diseases such as Alzheimer’s and Parkinson’s. By bridging mechanistic understanding and translational research, SOD1 PFFs accelerate the discovery of disease-modifying strategies for disorders driven by protein aggregation. |
| References |
1. Adachi T., et al. (1992). Clin. Chim. Acta. 212: 89-102. 2. Barrister J.V., et al. (1987). Crit. Rev. Biochem. 22:111-180. 3. Furukawa Y., O'Halloran T. (2006). Antioxidants & Redo Signaling. Vol 8, No 5,6. 4. Gao B., et al. (2003). Am J Physiol Lung Cell Mol Physiol 284: L917-L925. 5. Hassan H.M. (1988). Free Radical Biol. Med. 5: 377-385. 6. Kurobe N., et al. (1990) Biomedical Research. 11: 187-194 7. Wispe J.R., et al. (1989) BBA. 994: 30-36. 8. Xiao-Hong Liu., et al. (1993) Brain Research. 625: 29-37. 9. Furukawa Y., et al. (2013) FEBS 587(16): 2500-2505. |
























Reviews
There are no reviews yet.