Properties
| Storage Buffer | 1X PB pH7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau-441 (2N4R) Wild-Type Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-496) |
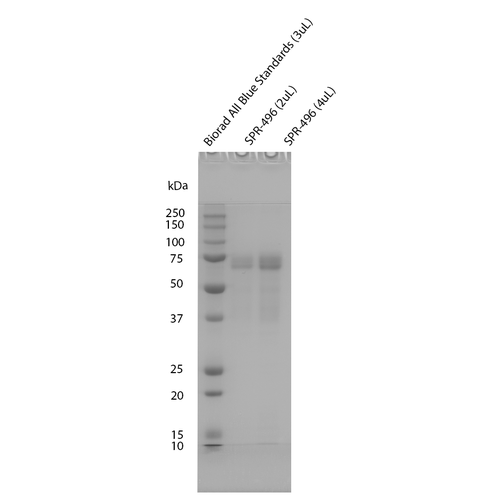
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-498 |
Biological Description
| Alternative Names | MAPT, Tau, Tau-441, 2N4R, Tau40, intracellular neurofibrillary tangles, NFTs, paired helical filaments, PHFs, microtubule-associated Tau, tubulin-associated unit, Paired Helical Filament-Tau, Phf-Tau, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1, Isoform 2 |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Tangles & Tau |
| Swiss Prot | P10636-8 |
| Scientific Background |
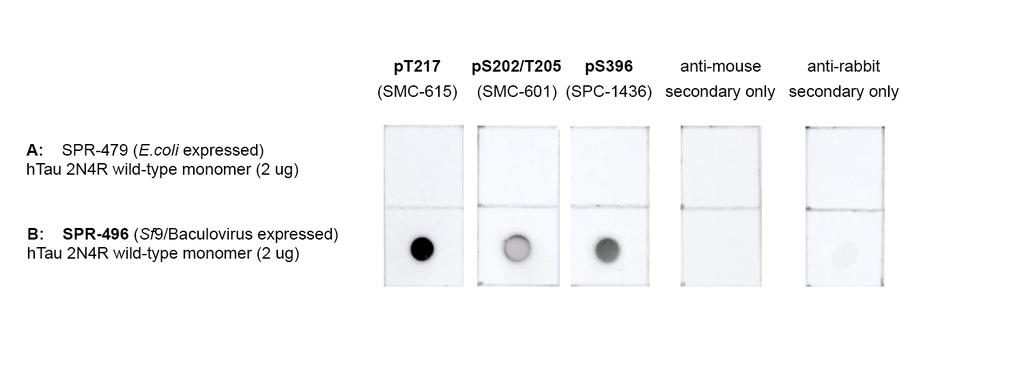
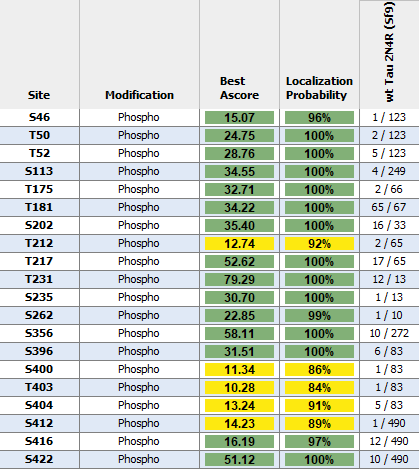
Tau protein, encoded by the MAPT gene (UniProt ID: P10636), is essential for stabilizing microtubules and maintaining neuronal architecture. The Tau-441 isoform, also known as 2N4R, is the longest variant expressed in the adult human brain, containing two N-terminal inserts and four microtubule-binding repeats. This isoform plays a central role in tauopathy research due to its structural complexity and physiological relevance. In its wild-type monomeric form, Tau-441 maintains proper microtubule dynamics and intracellular transport. However, under pathological conditions, even wild-type Tau can undergo conformational changes that lead to misfolding, hyperphosphorylation, and aggregation. These early molecular events are critical to understanding the onset of neurodegenerative diseases such as Alzheimer’s disease and frontotemporal dementia. Tau-441 monomers are widely used in experimental models to study the transition from functional protein to pathogenic species. Their behavior under stress, mutation, or post-translational modification provides insight into the mechanisms that drive Tau aggregation and neurotoxicity. These monomers also serve as a baseline for evaluating therapeutic strategies aimed at preserving Tau’s native state and preventing disease progression. By modeling the earliest stages of Tau dysfunction, Tau-441 wild-type monomers offer a robust platform for advancing biomarker discovery and the development of targeted interventions in neurodegenerative disease research. Hyper-phosphorylated tau can be generated via expression in the Sf9/Baculovirus system, with up to 20 sites confirmed by mass spectrometry and Western Blots with phospho-specific antibodies(3). Sf9/Baculovirus-expressed Tau 2N4R will readily form oligomers and fibrils in the absence of heparin. |
| References |
1. Goedert et al. 1989. Multiple Isoforms of Human Microtubule-associated Protein Tau: Sequences and Localization in Neurofibrilary Tangles of Alzheimer’s Disease. Neuron. doi: 10.1016/0896-6273(89)90210-9. 2. Iqbal K., Liu F., and Gong C.X. 2016. Tau and neurodegenerative disease: The story so far. Nat. Rev. Neurol. DOI: 10.1038/nrneurol.2015.225 3. Tepper et al. 2014. Oligomer Formation of Tau Protein Hyperphophorylated in Cells. The Journal of Biological Chemistry, DOI 10.1074/jbc.M114.611368 |






















Reviews
There are no reviews yet.