Properties
| Storage Buffer | 10 mM HEPES, 100 mM NaCl pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau-441 (2N4R) P301S Mutant Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-327) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-329 |
Biological Description
| Alternative Names | Tau-441, Tau-F, Tau 441, 2N4R, MAPT, TAU, MTBT1, MTBT2, MAPTL, PPND, PPP1R103, FTDP-17, PHF-Tau, Paired Helical Filament-Tau, Neurofibrillary Tangle, NFTs, intracellular neurofibrillary tangles, Tau aggregates, Tau inclusions, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1 |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Accession Number | NP_005901.2 |
| Gene ID | 4137 |
| Swiss Prot | P10636 |
| Scientific Background |
Tau-441, also known as the 2N4R isoform of the microtubule-associated protein tau (MAPT), is the longest isoform expressed in the adult human brain. It plays a critical role in stabilizing microtubules and maintaining neuronal structure and function. Under physiological conditions, tau monomers dynamically bind microtubules, supporting axonal transport and synaptic integrity. The P301S mutation, a proline-to-serine substitution at residue 301, is associated with familial frontotemporal dementia and other tauopathies. This mutation alters the conformation and biochemical properties of tau monomers, reducing their affinity for microtubules and increasing their susceptibility to misfolding and aggregation. P301S mutant monomers exhibit enhanced nucleation potential, facilitating the formation of toxic oligomers and fibrils that disrupt neuronal homeostasis. These misfolded tau species interfere with cytoskeletal dynamics, impair axonal transport, and activate neuroinflammatory pathways, contributing to progressive neurodegeneration. In transgenic models, P301S tau monomers initiate early pathological changes, including synaptic loss, mitochondrial dysfunction, and cognitive decline, even before the appearance of mature neurofibrillary tangles. Understanding the molecular behavior of P301S mutant tau monomers is essential for developing targeted therapies. Strategies aimed at stabilizing tau’s native conformation, inhibiting early aggregation events, or enhancing clearance mechanisms are under investigation to mitigate tau-driven neurodegeneration. Tau-441 P301S mutant monomers represent a critical early target in the cascade of tauopathy progression, offering valuable insights for therapeutic intervention and biomarker development. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatr. Psych.-Gerichtl. Med. 64, 146–148 (1907) 3. Matsumoto, G. et al. (2018). Int J Mol Sci. 19, 1497. 4. Goedert, M. and Spillantini, M. G. (2017). Mol Brain. 10:18. 5. Bugiani, O. et al. (1999). J Neuropathol Exp Neurol. 58(6):667-77. |
Product Images
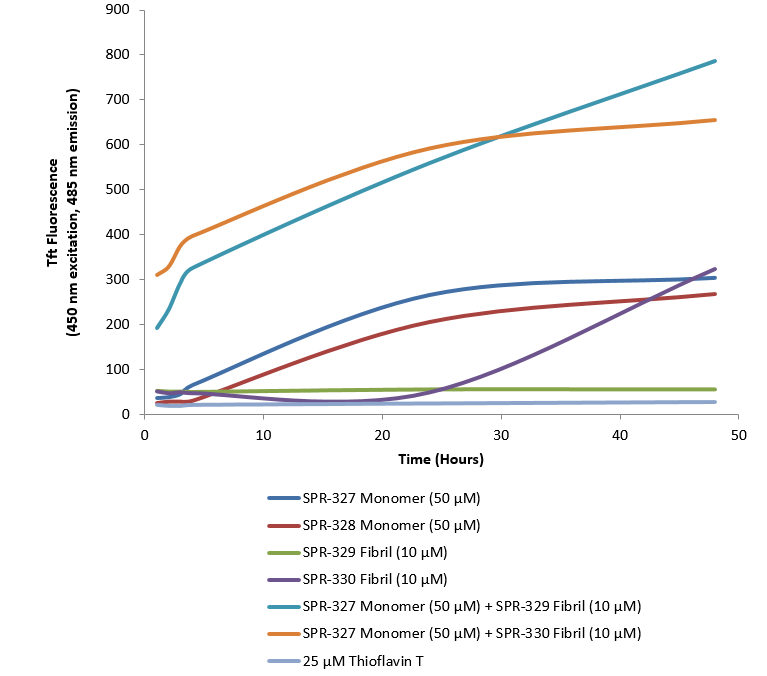
Thioflavin T is a fluorescent dye that binds to beta sheet-rich structures, such as those in tau fibrils. Upon binding, the emission spectrum of the dye experiences a red-shift and increased fluorescence intensity. Thioflavin T emission curves show increased fluorescence (correlated to tau aggregation) over time in tau monomers (SPR-327). A greater increase in fluorescence is seen when 50 µM monomer (SPR-327) is combined with 10 µM PFFs (SPR-329 or SPR-330), as the fibrils seed the formation of new fibrils from the pool of monomers. Thioflavin T ex = 450 nm, em = 485 nm. 10 uM heparin was added to each well. Note: We use molecular weight of 45.84 kDa for both tau monomer and fibril in calculations. We load 100µL/well for Thioflavin T assay so 50 µM is 229.2 µg/well and 10 µM is 45.84 µg/well.

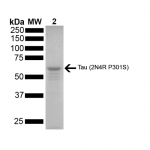





















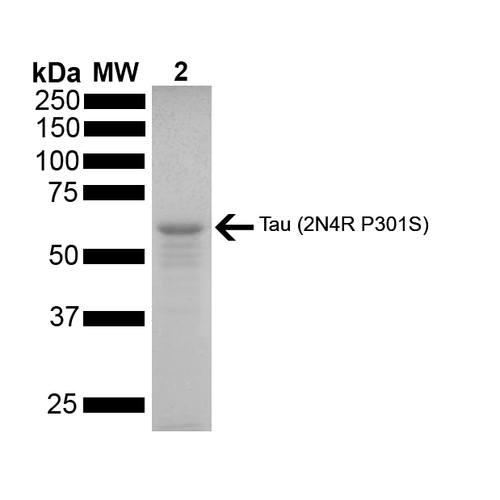
StressMarq Biosciences :
Based on validation through cited publications.