Properties
| Storage Buffer | PB pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau dGAE (297-391) Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-444) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-462 |
Biological Description
| Alternative Names | Tau dGAE, Tau 297–391, Truncated Tau, Tau fragment 297–391, microtubule-associated tau, MAPT, MAP, Tau-441, Paired Helical Filament-Tau, Phf-Tau, Neurofibrillary Tangle, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1, Isoform 2, tubulin-associated unit |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Accession Number | NP_005901.2 |
| Gene ID | 4137 |
| Swiss Prot | P10636 |
| Scientific Background |
Tau dGAE (297–391), a core fragment of the microtubule-associated protein tau, is gaining prominence in neurodegenerative disease research due to its intrinsic ability to self-assemble into filamentous aggregates. This monomeric fragment, derived from the C-terminal region of tau, represents a minimal structural unit capable of recapitulating disease-specific filament folds observed in conditions such as Alzheimer’s disease (AD) and chronic traumatic encephalopathy (CTE). Unlike full-length tau, dGAE monomers spontaneously aggregate in vitro, forming stable filaments that mimic pathological tau structures found in human brains. These filaments efficiently seed the aggregation of endogenous tau, triggering a cascade of misfolding events that culminate in neurofibrillary tangle formation—a hallmark of tauopathies. The dGAE fragment’s aggregation propensity and structural fidelity make it a powerful model for studying tau misfolding and for screening anti-aggregation therapeutics. Recent studies have demonstrated that dGAE-induced tau filaments propagate disease-specific conformations, suggesting a prion-like mechanism of tau spread. Moreover, targeting truncated tau species such as dGAE has shown promise in reducing tau pathology, restoring synaptic function, and improving cognitive outcomes in preclinical models. In summary, Tau dGAE (297–391) monomers serve as critical molecular initiators of tau aggregation and neurotoxicity. Their unique properties position them as valuable tools for mechanistic studies, biomarker development, and the design of targeted interventions in tau-driven neurodegenerative diseases. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatr. Psych.-Gerichtl. Med. 64, 146–148 (1907) 3. Al-Hilaly, Y.K. et al. Alzheimer's Disease-like Paired Helical Filament Assembly from Truncated Tau Protein Is Independent of Disulfide Crosslinking. J. Mol. Biol. 429(23):3650-3665 (2017) |
Product Images
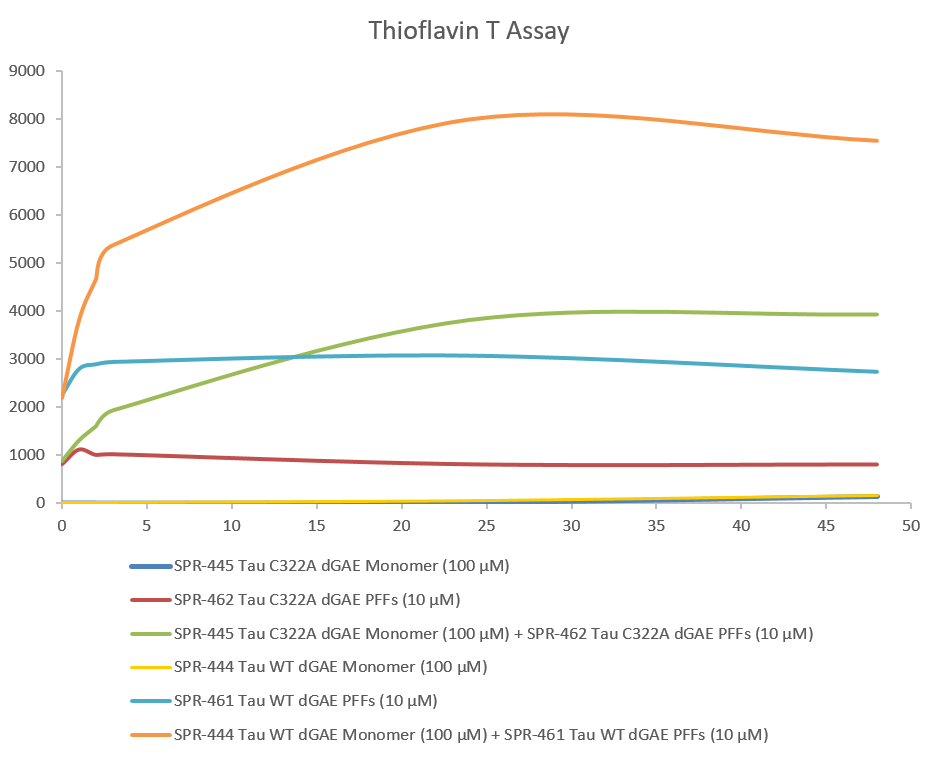
Thioflavin T is a fluorescent dye that binds to beta sheet-rich structures, such as those in tau fibrils. Upon binding, the emission spectrum of the dye experiences a red-shift and increased fluorescence intensity. Thioflavin T emission curves show increased fluorescence (correlated to tau aggregation) over time when truncated tau fragment (AA297-391) (dGAE) monomer is combined with truncated tau fragment (AA297-391) (dGAE) Pre-formed fibrils (Type 1).
























StressMarq Biosciences :
Based on validation through cited publications.