| Product Name | Tau dGAE (297-391) C322A Mutant Pre-formed Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
Human Recombinant Tau dGAE (297-391) C322A Mutant PFFs |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS-PAGE, In vivo assay, In vitro assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | 2 mg/mL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amino Acid Sequence | MIKHVPGGGSV QIVYKPVDLS KVTSKAGSLG NIHHKPGGGQ VEVKSEKLDF KDRVQSKIGS LDNITHVPGG GNKKIETHKL TFRENAKAKT DHGAE | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | Fragment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | 10.133 kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Activity | Thioflavin T emission curves show increased fluorescence (correlated to tau aggregation) over time when truncated tau fragment (AA297-391) (dGAE C322A) monomer is combined with truncated tau fragment (AA297-391) (dGAE C322A) Pre-formed Fibrils. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau dGAE (297-391) C322A Mutant Pre-formed Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-462) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Made with low endotoxin monomer. |
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-445. |
Biological Description
| Alternative Names | Tau dGAE C322A, Tau 297–391 C322A, Tau fragment 297–391 C322A, Truncated Tau C322A, microtubule-associated tau, MAPT, MAP, Tau-441, Paired Helical Filament-Tau, Phf-Tau, Neurofibrillary Tangle, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1, Isoform 2, tubulin-associated unit, 95-amino acid Tau fragment, Tau PFFs |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Accession Number | NP_005901.2 |
| Gene ID | 4137 |
| Swiss Prot | P10636 |
| Scientific Background |
Tau protein, encoded by the MAPT gene (UniProt ID: P10636), is essential for microtubule stabilization and neuronal function. In tauopathies such as Alzheimer’s disease, Tau undergoes pathological misfolding and aggregation, forming neurofibrillary tangles that disrupt neuronal integrity. The dGAE fragment (residues 297–391) represents the core region of Tau’s microtubule-binding domain and is highly prone to fibrillization. The C322A mutation within this fragment replaces cysteine with alanine at position 322, eliminating disulfide bond formation and enhancing fibril homogeneity. This modification yields structurally consistent pre-formed fibrils (PFFs), which are critical for reproducible studies of Tau aggregation and propagation. Tau dGAE C322A mutant PFFs closely mimic the β-sheet-rich architecture of pathological Tau aggregates found in human brains. Their stability and uniformity make them ideal for investigating the molecular underpinnings of Tau seeding, templated misfolding, and intercellular transmission—key processes in the progression of neurodegenerative diseases. These mutant fibrils are increasingly used in cellular and animal models to evaluate therapeutic interventions targeting Tau pathology. By enabling precise mechanistic studies and high-throughput screening, Tau dGAE C322A PFFs accelerate the development of disease-modifying treatments and biomarker discovery, reinforcing their value in translational neuroscience. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatr. Psych.-Gerichtl. Med. 64, 146–148 (1907) 3. Al-Hilaly, Y.K. et al. Alzheimer's Disease-like Paired Helical Filament Assembly from Truncated Tau Protein Is Independent of Disulfide Crosslinking. J. Mol. Biol. 429(23):3650-3665 (2017) |
Product Images
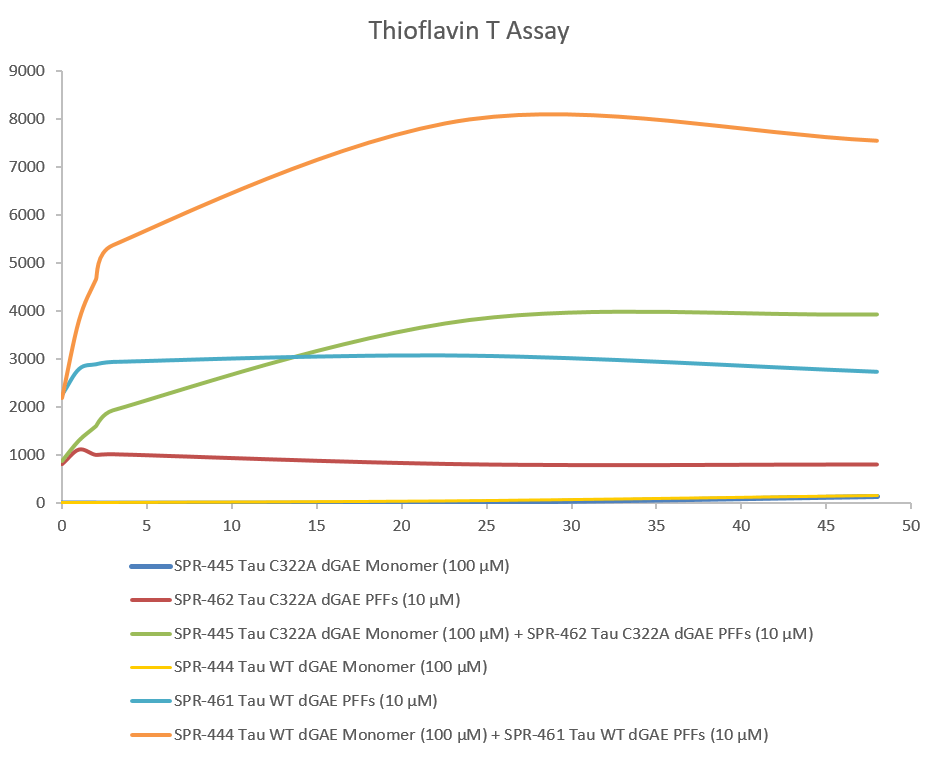
Thioflavin T is a fluorescent dye that binds to beta sheet-rich structures, such as those in tau fibrils. Upon binding, the emission spectrum of the dye experiences a red-shift and increased fluorescence intensity. Thioflavin T emission curves show increased fluorescence (correlated to tau aggregation) over time when truncated tau fragment (AA297-391) (dGAE C322A) monomer is combined with truncated tau fragment (AA297-391) (dGAE C322A) Pre-formed fibrils .

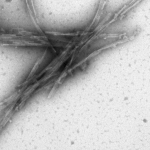






















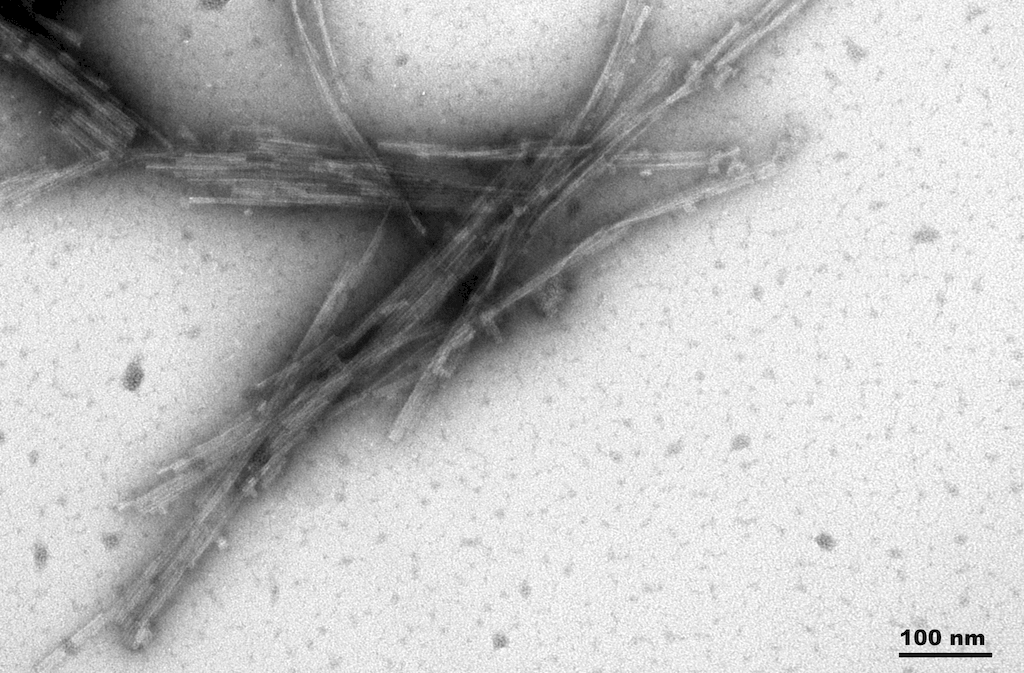
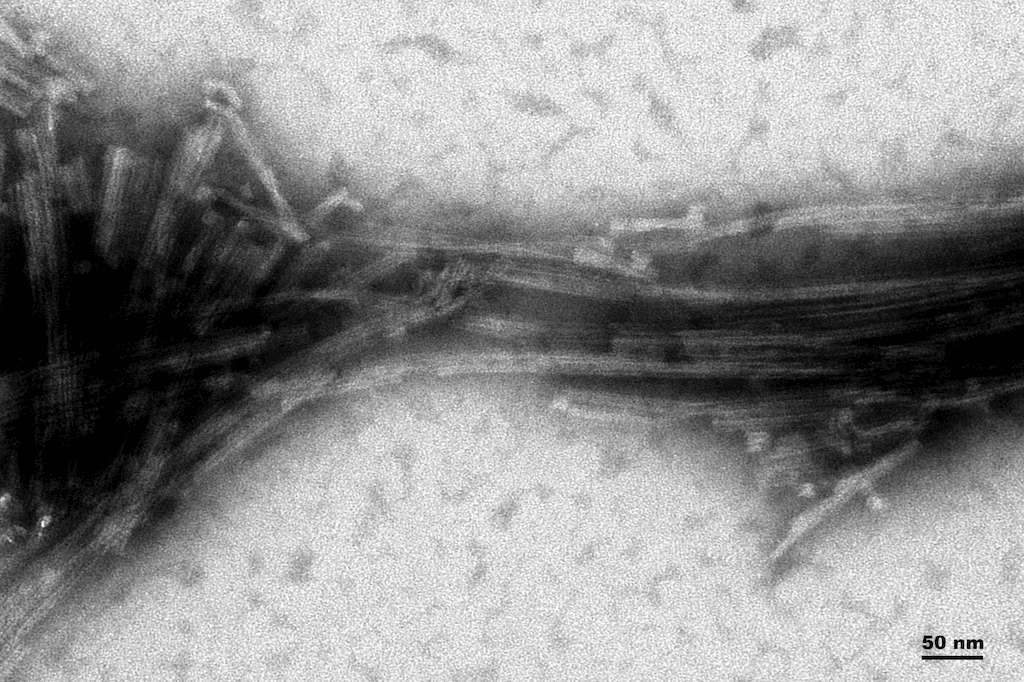
Reviews
There are no reviews yet.