Properties
| Storage Buffer | PBS pH 7.4, 5 mM DTT, 2 mM MgCl2 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau-441 (2N4R) P301S Mutant Filaments (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-463) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. |
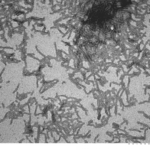
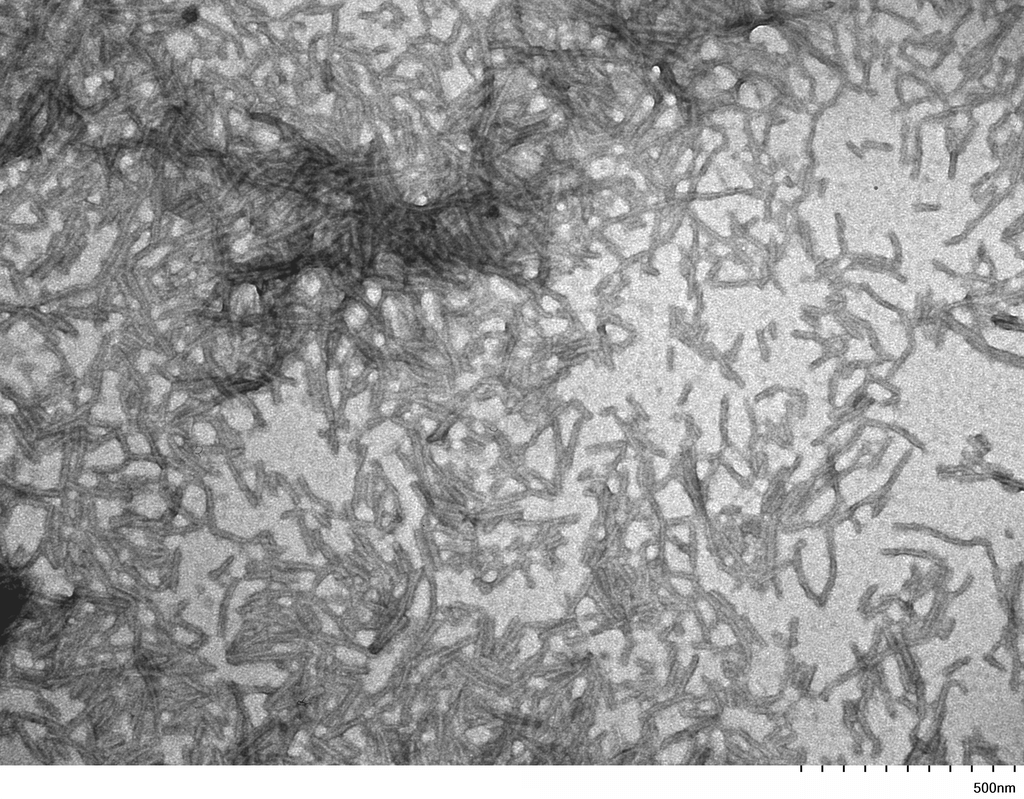
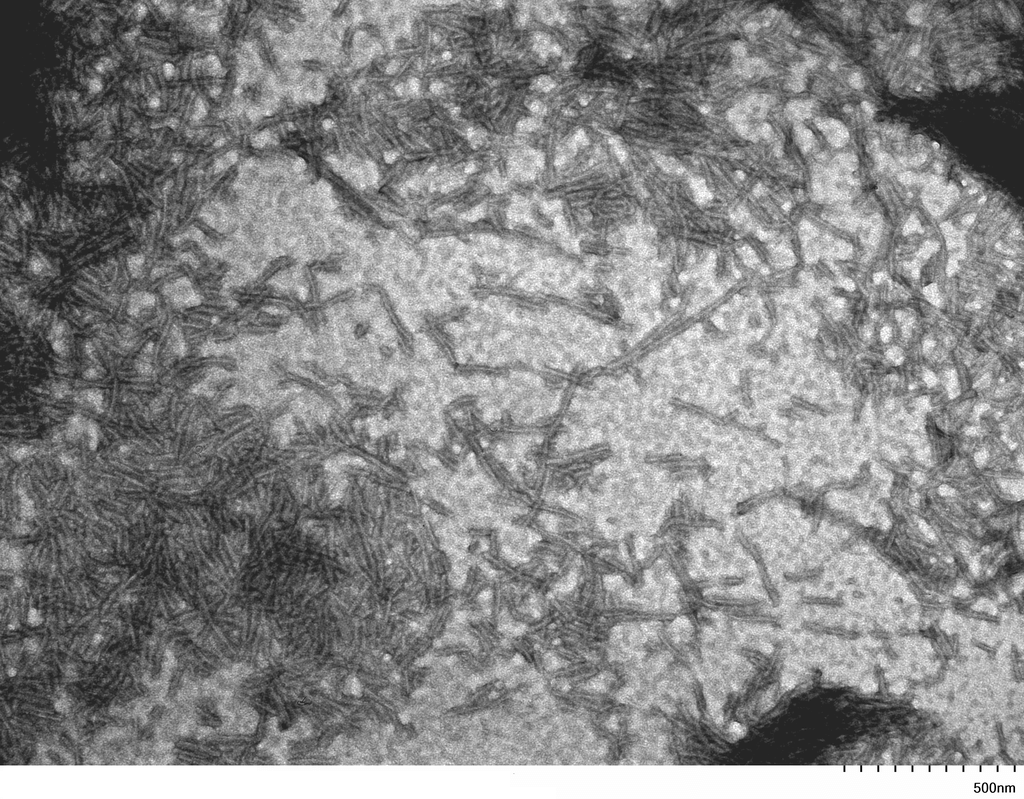
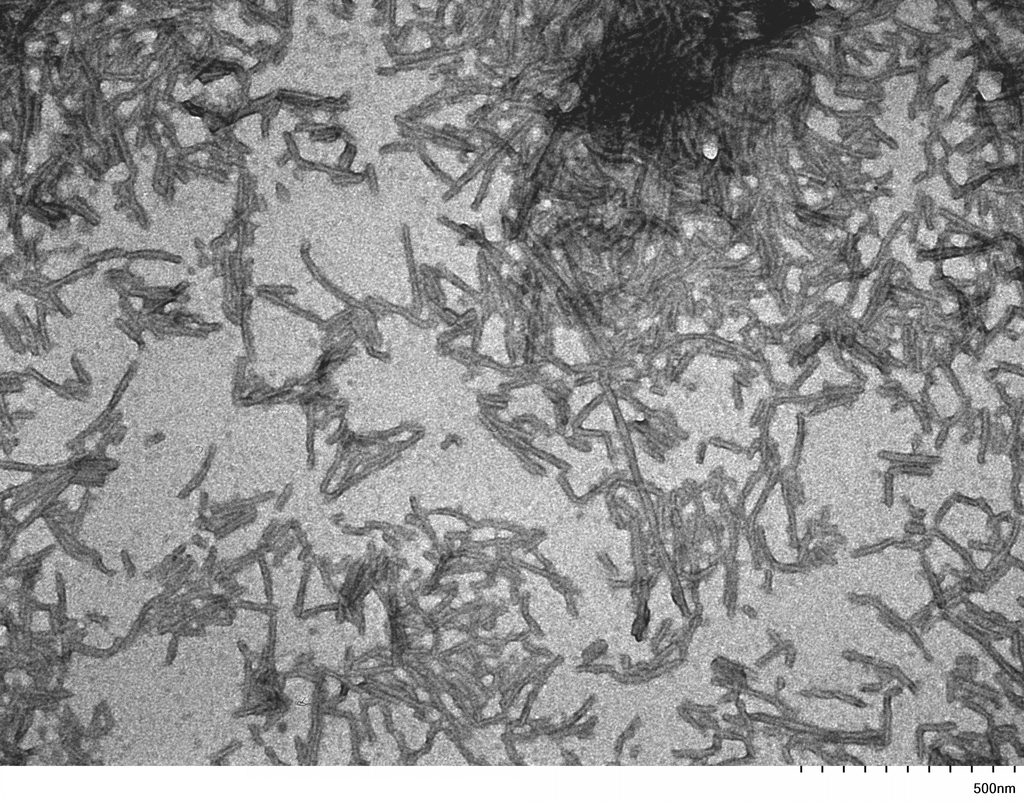
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-327. |
Biological Description
| Alternative Names | Tau-441, Tau-F, Tau 441, 2N4R, MAPT, TAU, MTBT1, MTBT2, MAPTL, PPND, PPP1R103, FTDP-17, PHF-Tau, Paired Helical Filament-Tau, Neurofibrillary Tangle, NFTs, intracellular neurofibrillary tangles, Tau aggregates, Tau inclusions, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1 |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Accession Number | NP_005901.2 |
| Gene ID | 4137 |
| Swiss Prot | P10636 |
| Scientific Background |
Tau protein, encoded by the MAPT gene (UniProt ID: P10636), is a microtubule-associated protein essential for maintaining neuronal structure and function. The full-length Tau-441 isoform (2N4R) contains two N-terminal inserts and four microtubule-binding repeats, representing the longest and most aggregation-prone variant expressed in the adult human brain. The P301S mutation, located within the microtubule-binding domain, significantly enhances Tau’s propensity to misfold and assemble into filamentous aggregates. These mutant filaments recapitulate key pathological features of tauopathies, including Alzheimer’s disease and frontotemporal dementia, where neurofibrillary tangles composed of hyperphosphorylated Tau disrupt neuronal integrity and function. Tau-441 P301S mutant filaments serve as a robust experimental model for studying the molecular mechanisms of Tau aggregation, seeding, and prion-like propagation. Their use in transgenic animal models and in vitro systems has advanced our understanding of Tau-induced neurotoxicity, synaptic dysfunction, and neuroinflammation. These filaments are also instrumental in preclinical drug development, enabling the screening of compounds that inhibit Tau aggregation, promote clearance, or block intercellular transmission. By closely mimicking human pathological Tau, Tau-441 P301S filaments provide a high-fidelity platform for biomarker discovery and therapeutic innovation in neurodegenerative disease research. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatr. Psych.-Gerichtl. Med. 64, 146–148 (1907) 3. Matsumoto, G. et al. (2018). Int J Mol Sci. 19, 1497. 4. Goedert, M. and Spillantini, M. G. (2017). Mol Brain. 10:18. 5. Guo, J. and Lee, M.Y. (2013). FEBS Lett. 587(6): 717-723. 6. Alberici, A. et al. (2004). J Neurol Neurosurg Psychiatry. 75:1607-1610. |





















Reviews
There are no reviews yet.