Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Transthyretin (TTR) Y78F Variant Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-452) |
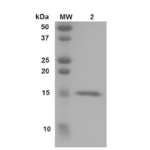
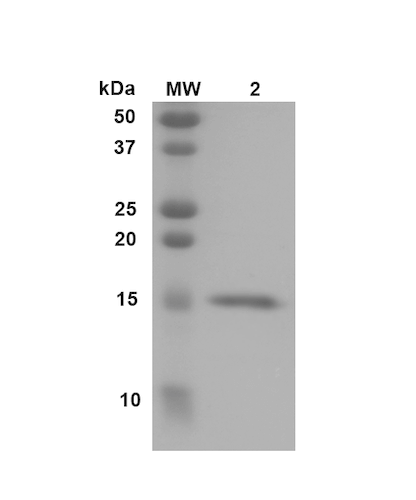
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-465 |
Biological Description
| Alternative Names | Transthyretin Y78F, TTR Y78F, ATTR, Amyloid polyneuropathy, Amyloidosis I, Carpal tunnel syndrome 1, CTS, CTS1, HEL111, HsT2651, PALB, Prealbumin, Prealbumin amyloidosis type I, Prealbumin Thyroxine-binding, TBPA, Thyroxine binding prealbumin, TTHY_HUMAN, TTR |
| Research Areas | ALS Disease, Alzheimer's Disease, Blood, Cardiovascular System, Cell Signaling, Lipid and lipoprotein Metabolism, Metabolism, Neurodegeneration, Neuroscience, Parkinson's Disease, Tangles & Tau |
| Cellular Localization | Cytoplasm, Extracellular exosome, Extracellular Region, Lysosome |
| Accession Number | NP_000362.1 |
| Gene ID | 7276 |
| Swiss Prot | P02766 |
| Scientific Background |
The Y78F variant of transthyretin (TTR), a key transport protein for thyroxine and retinol, is increasingly recognized for its role in neurodegenerative disease pathology. This single-point mutation destabilizes the native tetrameric structure of TTR, promoting dissociation into monomers that are prone to misfolding and aggregation. Unlike wild-type TTR, Y78F monomers exhibit enhanced amyloidogenic potential under physiological conditions. These misfolded species initiate the formation of toxic oligomers and fibrils, contributing to the extracellular deposition of amyloid in neural tissues. Such deposits are implicated in hereditary transthyretin amyloidosis (ATTR), particularly in forms affecting the central and peripheral nervous systems. Recent research has shown that Y78F monomers compromise the integrity of the blood–nerve barrier and trigger neuroinflammatory responses, exacerbating neuronal damage. Their structural instability and altered surface chemistry facilitate interactions with cellular membranes and immune cells, amplifying neurotoxicity. Understanding the biophysical properties and aggregation pathways of TTR Y78F monomers is essential for developing targeted therapies. Stabilizing the tetrameric form or inhibiting monomer misfolding offers promising strategies to prevent amyloid formation and mitigate neurodegeneration. In summary, TTR Y78F variant monomers are critical molecular drivers of amyloid pathology and neuroinflammation. Their study provides valuable insights into the mechanisms of neurodegenerative diseases and informs the development of precision therapeutics for ATTR and related disorders. |
| References |
1. Zeldenrust S.R., Benson M.D. (2010). Wiley. pp. 795–815. 2. Westermark P., Sletten K., Johansson B., Cornwell G.G. (1990). Proc. Natl. Acad. Sci. U.S.A. 87(7): 2843–5. 3. Andrade C. (1952). Brain. 75(3): 408–27. 4. Coelho T. (1996). Curr. Opin. Neurol. 9(5): 355–9. 5. Jacobson D.R, et. al. (1997). N. Engl. J. Med. 336(7): 466–73. 6. Li X. (2011). Mol Neurodegener. 6(1):79. 7. Terazaki H.,m et al. (2006) Lab Invest. 86(1): 23-31. |





















Reviews
There are no reviews yet.