| Product Name | Xanomeline Tartrate |
| Description |
Selective muscarinic receptor agonist |
| Purity | >98% (HPLC); NMR (conforms) |
| CAS No. | 152854-19-8 |
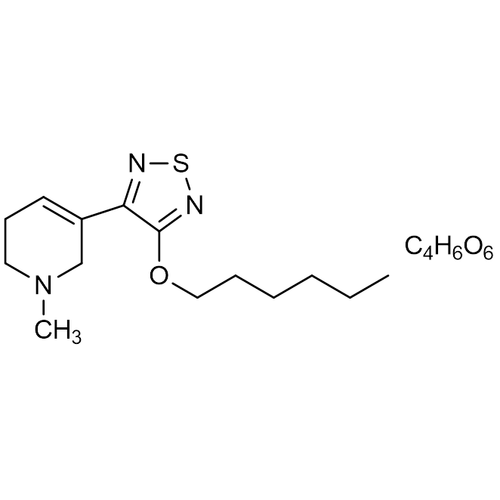
| Molecular Formula | C14H23N3OS • C4H6O6 |
| Molecular Weight | 431.5 |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | -20ºC |
| Shipping Temperature | Shipped Ambient |
| Product Type | Agonist |
| Solubility | May be dissolved in DMSO (45 mg/ml); or water (70 mg/ml) |
| Source | Synthetic |
| Appearance | Off-white powder |
| SMILES | CCCCCCOC1=NSN=C1C2=CCCN(C2)C.[C@@H]([C@H](C(=O)O)O)(C(=O)O)O |
| InChI | InChI=1S/C14H23N3OS.C4H6O6/c1-3-4-5-6-10-18-14-13(15-19-16-14)12-8-7-9-17(2)11-12;5-1(3(7)8)2(6)4(9)10/h8H,3-7,9-11H2,1-2H3;1-2,5-6H,(H,7,8)(H,9,10)/t;1-,2-/m.1/s1 |
| InChIKey | SJSVWTMVMBGIHQ-LREBCSMRSA-N |
| Safety Phrases |
Classification: Warning Hazard statement(s): H302 (100%): Harmful if swallowed Precautionary statement(s): P264, P270, P301+P317, P330, and P501 |
| Cite This Product | Xanomeline Tartrate (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SIH-642) |
Biological Description
| Alternative Names | 3-[4-(Hexyloxy)-1,2,5-thiadiazol-3-yl]-1,2,5,6-tetrahydro-1-methylpyridine, tartrate, LY-246708; NNC-11-0232 |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience |
| PubChem ID | 71456 |
| Scientific Background |
A functionally selective M1 muscarinic receptor agonist with high affinity for central muscarinic subtypes (EC₅₀ = 0.3, 92.5, 5, 52, and 42 nM for M1, M2, M3, M4, and M5, respectively). Its strong selectivity for M1 receptors underlies a range of central nervous system effects, including positive cognitive and behavioral improvements in models of schizophrenia and Alzheimer’s disease. Beyond its neurocognitive actions, the compound also suppresses pro‑inflammatory cytokine responses and enhances survival in sepsis models. Additionally, it demonstrates potent analgesic efficacy in rodent models of chronic inflammatory and neuropathic pain, indicating broad therapeutic potential across neuroimmune and pain‑processing pathways. |
| References |
1.,JN Heinrich et al. Eur. J. Pharmacol. 2009 605:53 2.,J Jakubik et al. Physiol. Res. 2008 57 Suppl 3:S39 3.,AM Bender et al. ACS Chem. Neurosci. 2017 8:435 4.,M Rosas-Ballina et al. Brain Behav. Immun. 2015 44:19 5.,G Martino et al. Pain 2011 152:2852 |

Reviews
There are no reviews yet.