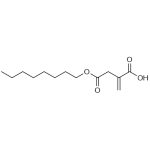
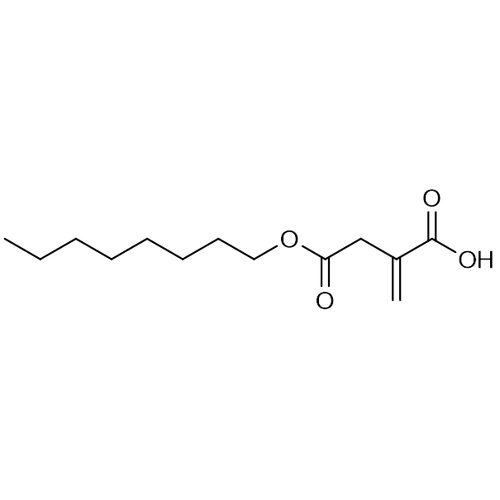
| Product Name | 4-Octyl itaconate |
| Description |
Neuroprotective Agent |
| Purity | >98% (TLC); NMR (Conforms) |
| CAS No. | 3133-16-2 |
| Molecular Formula | C13H22O4 |
| Molecular Weight | 242.3 |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | -20ºC |
| Shipping Temperature | Shipped Ambient |
| Product Type | Inhibitor |
| Solubility | May be dissolved in DMSO (25 mg/ml) |
| Source | Snythetic |
| Appearance | White to off-white powder |
| SMILES | CCCCC(CCC)OC(=O)C(=C)CC(=O)[O-] |
| InChI | InChI=1S/C13H22O4/c1-4-6-8-11(7-5-2)17-13(16)10(3)9-12(14)15/h11H,3-9H2,1-2H3,(H,14,15)/p-1 |
| InChIKey | GIRJEIMINMHXQS-UHFFFAOYSA-M |
| Safety Phrases |
Classification: Warning. Hazard statement(s): H302 Harmful if swallowed H315 Causes skin irritation H319 Causes serious eye irritation H335 May cause respiratory irritation Precautionary statement(s): P261 Avoid breathing dust/fume/gas/mist/vapours/spray. P264 Wash hands thoroughly after handling P270 Do not eat, drink or smoke when using this product. P271 Use only outdoors or in a well-ventilated area. P280 Wear protective gloves/protective clothing/eye protection/face protection |
| Cite This Product | 4-Octyl itaconate (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SIH-637) |
Biological Description
| Alternative Names | 4-Octyl methylenesuccinate; or n-Octyl 3-carboxy-3-butenoate |
| Research Areas | Atherosclerosis, Cardiovascular System, Immunology, Neuroscience, Oxidative Stress |
| PubChem ID | 71360202 |
| Scientific Background | 4‑Octyl itaconate is a cell‑permeable derivative of itaconic acid that activates the neuroprotective transcription factor Nrf2 through alkylation of KEAP1¹. In neural systems, Nrf2 activation is closely associated with antioxidant defense and suppression of neuroinflammatory signaling. 4‑Octyl itaconate inhibits several regulated cell‑death pathways relevant to neurological disease, including ferroptosis²—a major contributor to neuronal and glial oxidative injury—as well as gasdermin B–mediated pyroptosis³, a key driver of neuroinflammatory cytokine release. In addition, it enhances secretion of anti‑inflammatory mediators⁴ and promotes a wound‑resolving, pro‑repair phenotype in activated macrophages⁵, suggesting potential to shift microglia and infiltrating myeloid cells toward a neuroprotective state in models of neurodegeneration or CNS injury. |
| References |
1. EL Mills et al. Nature 2018 556:113 2. X Pan et al. Int. Immunopharmacol. 2024 137:112531 3. W Gong et al. Cell Prolif. 2024 57:e13711 (Epub) 4. C Diskin et al. J. Immunol. 2023 211:1032 5. S Maassen et al. Redox. Biol. 2023 59:102591 6. L Zhang, et al. Inflammation. 2025 49(1): https://pubmed.ncbi.nlm.nih.gov/41405773/ |

Reviews
There are no reviews yet.