| Product Name | Compound E |
| Description |
Gamma secretase Inhibitor |
| Purity | ≥98% (TLC); NMR (Conforms) |
| CAS No. | 209986-17-4 |
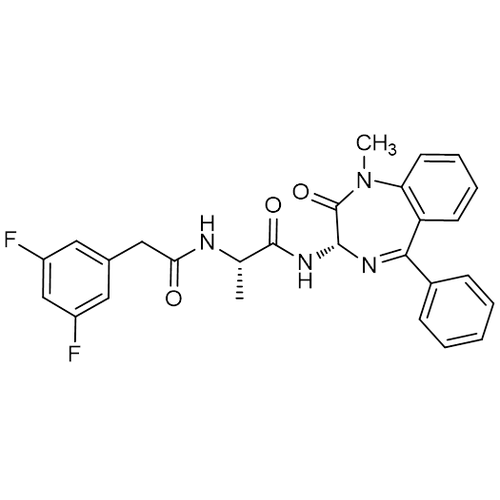
| Molecular Formula | C27H24F2N4O3 |
| Molecular Weight | 490.5 |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | -20ºC |
| Shipping Temperature | Shipped Ambient |
| Product Type | Neuronal Inducer |
| Solubility | May be dissolved in DMSO (25 mg/ml) |
| Source | Synthetic |
| Appearance | White powder |
| SMILES | C[C@@H](C(=O)N[C@@H]1C(=O)N(C2=CC=CC=C2C(=N1)C3=CC=CC=C3)C)NC(=O)CC4=CC(=CC(=C4)F)F |
| InChI | InChI=1S/C27H24F2N4O3/c1-16(30-23(34)14-17-12-19(28)15-20(29)13-17)26(35)32-25-27(36)33(2)22-11-7-6-10-21(22)24(31-25)18-8-4-3-5-9-18/h3-13,15-16,25H,14H2,1-2H3,(H,30,34)(H,32,35)/t16-,25+/m0/s1 |
| InChIKey | JNGZXGGOCLZBFB-IVCQMTBJSA-N |
| Safety Phrases | Not a hazardous substance or mixture. |
| Cite This Product | Compound E (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SIH-640) |
Biological Description
| Alternative Names | N-[(1S)-2-[[(3S)-2,3-Dihydro-1-methyl-2-oxo-5-phenyl-1H-1,4-benzodiazepin-3-yl]amino]-1-methyl-2-oxoethyl]-3,5-difluorobenzeneacetamide |
| Research Areas | Cancer, Neurodegeneration, Neuroscience, Stem Cells |
| PubChem ID | 11306390 |
| Scientific Background |
A highly potent, selective, and cell‑permeable γ‑secretase inhibitor that blocks the proteolytic processing of both amyloid precursor protein (APP) and Notch, with IC₅₀ values of ~3 nM¹˒². By inhibiting γ‑secretase–mediated cleavage of APP, the compound robustly suppresses amyloid‑β (Aβ) production, a key pathological driver in Alzheimer’s disease⁵. In neural systems, this inhibitor demonstrates broad neuroscience‑relevant activity: it reduces proliferation and promotes differentiation of neuroblastoma cells³, and it induces neuronal differentiation from stem cells⁴, making it a valuable tool for studying neurodevelopment, neural lineage specification, and disease‑associated signaling pathways. |
| References |
1. D. Beher et al., J. Biol. Chem., 2001, 276:45394 2. G. Zhao et al., J. Biol. Chem., 2004, 279:50647 3. G. Ferrari‑Toninelli et al., Neuro‑Oncol., 2010, 12:1231 4. W. Li et al., Proc. Natl. Acad. Sci. USA., 2011, 108:8299 5. |


Reviews
There are no reviews yet.