PLGA Nanoparticles Attenuate Tau Aggregation
The microtubule-associated protein tau plays a critical role in maintaining the structure and stability of neurons. It is involved in several neurodegenerative disorders collectively known as tauopathies, including Alzheimer’s disease (AD), frontotemporal dementia (FTD), and chronic traumatic encephalopathy (CTE). The progressive decline in motor and cognitive skills observed in affected individuals is primarily driven by the hyperphosphorylation and aggregation of tau, which leads to the formation of neurofibrillary tangles (NFTs) that extensively damage neurons over time. Furthermore, environmental factors such as temperature and pH have recently been shown to be able to influence tau aggregation dynamics. As pathological tau accumulation is central to disease progression, researchers are actively exploring strategies to delay or prevent tau aggregation, in order to effectively treat neurodegeneration.
Elucidating environmental influences
In a recent investigation, scientists at the University of Alberta, Canada, utilized FDA-approved poly(D,L-lactide-co-glycolic acid) (PLGA) nanoparticles to explore how both nanoparticles and temperature influence tau aggregation. Their unique approach incorporated an array of biophysical, biochemical, and structural techniques. While nanoparticles are commonly employed in medicine as delivery systems for therapeutic agents, due to their capacity to traverse the blood-brain barrier, this particular study by Paul et al. employed native nanoparticles, meaning they were not loaded with any active drug. Prior studies have indicated that such nanoparticles can suppress amyloid beta aggregation in animal models. Building on this, the researchers examined how temperature affects the aggregation behavior of various tau isoforms in vitro.
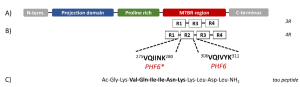
Tau isoforms arise through alternative splicing of the microtubule-associated protein tau (MAPT) gene located on chromosome 17. In the adult human brain, six distinct tau isoforms are expressed. Splicing at the N-terminal region yields isoforms with zero, one, or two N-terminal inserts (0N, 1N, or 2N), while additional splicing within the microtubule-binding domain produces isoforms containing either three or four repeat regions (3R or 4R). Within this domain, two short peptide sequences, PHF6 and PHF6*, have been identified as critical drivers of tau aggregation.
Figure 1: Schematic structure of tau isoforms. (A) The tau protein domains: N-terminal projection domain, proline-rich region, microtubule-binding region (MTBR) and C-terminal domain. (B) The MTBR may contain three (3R) or four (4R) repeat units (R1 to R4) separated by short inter-repeat regions containing the PHF6 and PHF6* regions. (Figure taken from Honish, C. et al, 2022 used under license CC BY 4.0).
In vitro analysis of tau aggregation dynamics using StressMarq’s neurodegenerative protein constructs
Subsequently, Paul et al. utilized a recombinant in-house preparation of the 0N4R tau isoform, alongside StressMarq’s commercial Tau-441 (2N4R) Wild-Type Monomers (catalog# SPR-479), to conduct a series of in vitro tau aggregation experiments. Aggregation was initiated using heparin across a temperature range of 27°C to 40°C and tau concentrations from 2.5 μM to 20 μM. Over a 40-hour period, the kinetics of tau aggregation were tracked using a Thioflavin T (ThT) fluorescence assay. ThT is a dye that fluoresces upon binding to the cross-beta sheet structures characteristic of tau fibrils.
Interestingly, the researchers observed that higher concentrations of tau 0N4R led to increased aggregation. Additionally, elevated temperatures shortened the lag phase preceding fibril formation and accelerated the aggregation rate across all tested concentrations. In parallel experiments, tau 2N4R aggregation at a fixed temperature of 37°C also showed a dose-dependent increase. Lastly, these findings were corroborated by scanning transmission electron microscopy (STEM), which revealed progressively larger aggregates with rising temperature and protein concentration.
The role of nanoparticles
Further experiments were conducted to evaluate the impact of PLGA nanoparticles at concentrations of 5, 10, and 20 μM on tau aggregation kinetics. The nanoparticles demonstrated a dose-dependent inhibitory effect on the aggregation of 10 μM 0N4R tau across all tested temperatures. As PLGA concentration increased, the lag phase preceding fibril formation was extended, and the overall rate of fibril growth was reduced.
STEM revealed that the nanoparticles were binding directly to tau fibrils, inducing a conformational shift that became more pronounced at elevated temperatures. In addition, a bicinchoninic acid (BCA) protein assay confirmed that soluble tau levels rose in correlation with increasing PLGA concentrations. Similar trends were observed with the 2N4R isoform. Although this assay was conducted at a single temperature (37°C), the inhibitory effect of PLGA was even more pronounced at the highest concentration (20 μM) compared to 0N4R.
Disassembly of tau aggregates
To assess whether PLGA could actively disassemble pre-formed tau fibrils, 0N4R aggregates were incubated at various temperatures with and without PLGA. ThT fluorescence assays confirmed that the nanoparticles facilitated fibril disassembly, with the rate of breakdown increasing alongside temperature. The researchers further explored PLGA’s interaction with different tau conformers—monomers, oligomers, and fibrils—by incubating 0N4R preparations with or without 20 μM PLGA. They found that the nanoparticles reduced antibody binding at specific sites on oligomers and fibrils. Analysis of the blocked epitopes suggested that PLGA interacts primarily with the microtubule-binding region (MTBR) and the C-terminal domain. Notably, the MTBR contains the PHF6 and PHF6* motifs, which are critical to tau aggregation.
Summary
Using various in vitro experimental techniques, the scientists demonstrated that heparin-induced aggregation of tau isoform 0N4R increases in a concentration-dependent manner as the temperature rises from 27°C to 40°C. Notably, native PLGA nanoparticles not only mitigated tau aggregation but also facilitated the disassembly of mature tau fibrils. Evidence suggests that these nanoparticles exert their inhibitory effect by interacting with the MTBR and the C-terminal domain of the tau protein. The inclusion of tau isoform 2N4R in several assays indicates that the observed effects are not isoform-specific. Given prior findings that temperature plays a significant role in tau regulation, the authors concluded that native PLGA nanoparticles may hold therapeutic promise for treating tau-related neurodegenerative disorders.
Related StressMarq Products
StressMarq Biosciences proudly offers a diverse selection of high-quality reagents to support neurodegenerative disease research, including monomeric, oligomeric, and fibrillar tau protein constructs. In addition, StressMarq’s unique selection of tau antibodies, such as the human tau 2N isoform-specific Tau 2N (2N3R/2N4R) Antibody (catalog# SPC-813), supports a wide range of research applications. Visit our website for more information, including the latest scientific publications using our specialized tau, amyloid beta and alpha synuclein proteins.
References
- Temperature-dependent aggregation of tau protein is attenuated by native PLGA nanoparticles under in vitro conditions. Paul, P.S. et al., Int J Nanomedicine. 2025.
- Effect of trehalose and ceftriaxone on the stability of aggregating-prone peptide containing PHF6* sequence: An SRCD study. Honish, C. et al., Intl J Mol Sci. 2022.


Leave a Reply