Understanding Parkinson’s: Diagnostics, Therapies, & History
Parkinson’s disease (PD) is a neurodegenerative disease caused by the death of dopaminergic (dopamine-producing) neurons in the brain. The subsequent decrease of dopamine in the brain causes a range of movement and motor symptoms, primarily bradykinesia, involuntary tremors, and rigidity. Parkinson’s disease is the second most prominent neurodegenerative disease globally (behind Alzheimer’s disease). Though PD has been clinically recognized for over 200 years, there is no cure and limited treatment options.
Parkinson’s disease was first clinically described by James Parkinson, an English surgeon, in 1817. However, written descriptions matching the symptoms of PD can be found dating back to 1000 BC. Dr. Parkinson published his findings in an 1817 essay titled ‘An Essay on the Shaking Palsy’. This essay followed six case studies and was the first medical document to describe the disease. Shortly after, the French doctor Jean-Martin Charcot used James Parkinson’s findings to establish a more rigorous diagnostic criteria and description of PD, as well as officially naming it Parkinson’s disease to honour James Parkinson’s initial findings.
Synucleinopathies
Over one hundred years after James Parkinson’s description of PD, the disease would be categorized as a synucleinopathy, a category of neurodegenerative diseases defined by the accumulation and misfolding of alpha synuclein protein in the brain. Synucleinopathies encompass diseases such as Parkinson’s disease, dementia with Lewy bodies, and multiple systems atrophy (MSA), though each of these diseases has unique mechanisms of action, symptoms, and treatment options. Under normal conditions, alpha synuclein is implicated in the release of neurotransmitters, and it is not entirely clear what causes this transition from normality to the formation of neurotoxic aggregations.
Parkinson’s disease cases are divided into either familial variants or sporadic variants. Familial PD is caused by high-penetrance genetic variants. As the name suggests, these genetic variants are often passed directly from parent to child. However, often the disease onset is not until much later in life.
Conversely, sporadic PD does not have one clear cause and is often due to a combination of risk factors such as aging, environmental factors, and genetic mutations. Approximately 90-95% of all cases of Parkinson’s disease are sporadic. Because of this, the timeline for diagnosing PD is highly reliant upon the development of hallmark motor symptoms. However, these motor symptoms often occur years after the initial onset of the changes in the brain that cause disease. Like other neurodegenerative diseases, aging is the greatest risk factor for Parkinson’s and the disease is most typically diagnosed in those older than 60 years of age.
Diagnostics
The main challenge with diagnosing Parkinson’s disease is the delay in the manifestation of physical symptoms compared to the disease progression. As an example, the physical tremors that often lead to PD diagnosis may not manifest until dopaminergic neuron functioning has been reduced by over 50%. Additionally, sporadic or familial Parkinson’s disease is often not visible when using radiographic methods (e.g. CT, MRI, PET scans). Although these scans can help rule out other neurodegenerative diseases in diagnosing PD, they are often not used until physical symptoms develop.
There has been a concerted effort to develop diagnostic tools that can accurately identify Parkinson’s before the onset of symptoms, though results are still limited. One such tool is the use of biomarkers, which is a characteristic indicator of a specific biological process – often found in the blood or other biological fluid. However, there is significant overlap between biomarkers for PD and other neurodegenerative diseases. As such, these tests do not currently provide conclusive diagnostic results.
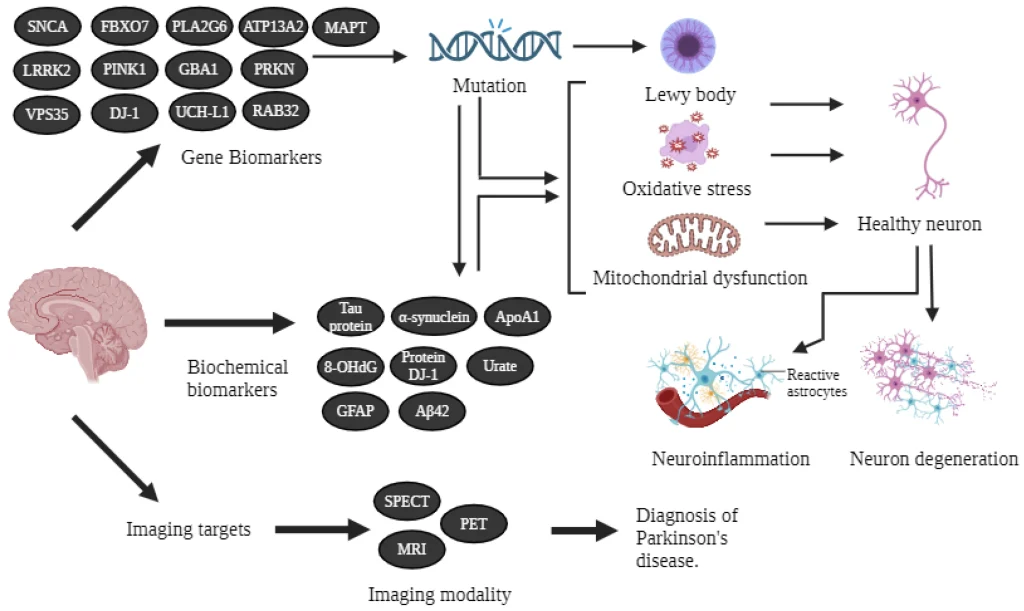
Figure 1. Schematic representation of PD biomarkers from Arya et al., 2024.
Therapeutics
As there is no cure for Parkinson’s disease, therapeutics that can slow the progression of symptoms have been crucial for patient care. The gold standard for pharmacologic treatment of PD symptoms has been the drug Levodopa. Levodopa is a dopamine precursor and has been used as a treatment for motor symptoms since the mid-1960s. In later-stage treatments, Levodopa is often combined with Carbidopa, a decarboxylase inhibitor. This combination allows for a greater bioavailability of Levodopa in patients with severely decreased dopamine production. However, Levodopa has a range of side effects which prohibit its widespread use, and such dopaminergic drugs may become less effective as the disease progresses.
Aside from Levodopa, there are a variety of drugs approved to treat different aspects of PD symptoms. These include, but are not limited to anticholinergics, COMT inhibitors, MAO-B inhibitors, NMDA receptor inhibitors, and various dopamine agonists for early-stage treatment. Outside of pharmacological interventions, stretching, strengthening, and voice training has been used in early-stage treatment to aid with balance, motor skills, and voice disorders. Though, the clinical evidence does not provide a conclusive verdict on how effective these interventions are.
Research is ongoing into new therapeutic approaches for both Parkinson’s disease and synucleinopathies. A recent innovative study published in the journal Molecular Therapy used StressMarq’s Mouse Alpha Synuclein Pre-formed Fibrils and Monomers (catalog# SPR-324 and SPR-323) to investigate the use of AAV-mediated artificial miRNA to block the spread of pathogenic alpha synuclein. Though this research is still in the early stages, continued innovation in therapeutics is critical.
Summary
Parkinson’s disease is a complex neurodegenerative disease whose onset is unpredictable, and patients are often diagnosed late in the disease progression. As the incidence and prevalence of Parkinson’s disease continues to rise, sustained research initiatives focused on advancing mechanistic understanding and enabling earlier diagnosis are being pursued with increasing urgency. Particularly, given that clinical diagnosis typically occurs after substantial disease progression, emphasis on early diagnosis and interventions are crucial.
Thanks in part to the relentless work of nonprofits such as the Michael J. Fox Foundation, awareness and funding for this research has grown vastly in the last 30 years. This includes a diverse array of research into the causes, progression, diagnostics, and therapeutics to improve outcomes and quality of life for those diagnosed with Parkinson’s.
Related StressMarq Products
Continued research into understanding disease mechanisms, identifying therapeutic targets, and developing diagnostics for Parkinson’s disease is crucial. To support this cutting-edge research, StressMarq Biosciences manufactures a wide range of specialized proteins for modelling neurodegenerative diseases such as Parkinson’s. These preparations include monomeric, oligomeric and fibrillar alpha synuclein, tau, amyloid beta, SOD1, TDP-43, and TTR for disease modelling, mechanistic studies, and pre-clinical drug discovery. To learn more about how StressMarq products are being used to drive advances in neurodegeneration research, visit our product citations hub page.
References
- Alzheimer’s and Parkinson’s disease therapies in the clinic. Chopade et al., BioTM. 2022
- Artificial miRNA-mediated reduction of SNCA for the treatment of α-synucleinopathies. Elmer et al., Mol. Ther. 2026
- Current Treatments and New, Tentative Therapies for Parkinson’s Disease. Pardo-Moreno et al., Pharmaceutics. 2023
- Parkinson’s Disease. Halli-Tierney et al., Am Fam Physician. 2020
- Parkinson Disease. 2023
- Parkinson’s 101. 2026
- Parkinson’s Disease: Biomarkers for Diagnosis and Disease Progression. Arya et al., Int. J. Mol. Sci. 2024
- The History of Parkinson’s Disease: Early Clinical Descriptions and Neurological Therapies. Christopher G Goetz. Cold Spring Harb Perspect Med. 2011


Leave a Reply