Properties
| Storage Buffer | 1X PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange & SEC purified |
| Cite This Product | Human Recombinant Alpha Synuclein E114C Mutant Monomers: ATTO 488 (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-517-A488) |
| Certificate of Analysis | Protein certified >95% pure on SDS-page and nanodrop analysis, endotoxin below 5 EU/mL at 2 mg/mL |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-518-A488 |
Biological Description
| Alternative Names | Alpha Synuclein E114C, Alpha-synuclein, Alpha synuclein, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, PD1, PARK4, Synuclein Alpha, Asyn, ATTO labelled Alpha Synuclein monomers |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Swiss Prot | P37840 |
| Scientific Background |
Alpha-synuclein (α-synuclein), encoded by the SNCA gene, is a presynaptic neuronal protein involved in synaptic vesicle trafficking, neurotransmitter release, and SNARE-complex assembly. Misfolding and aggregation of α-synuclein are central to the pathology of Parkinson’s disease and other synucleinopathies. The E114C mutation introduces a cysteine residue at position 114, enabling site-specific labeling and cross-linking for advanced biochemical and biophysical studies. This engineered variant retains the core functional domains of wild-type α-synuclein while offering enhanced experimental versatility. E114C mutant monomers are particularly useful for probing conformational changes, oligomerization pathways, and membrane interactions under physiological and pathological conditions. In neurodegenerative disease research, E114C monomers facilitate high-resolution analysis of early aggregation events and protein-protein interactions. Their compatibility with thiol-reactive probes allows for precise tracking of α-synuclein dynamics in vitro and in vivo. These monomers are instrumental in identifying molecular triggers of misfolding, mapping interaction networks, and screening therapeutic compounds that stabilize native conformations or prevent toxic oligomer formation. By enabling targeted investigation of α-synuclein structure and behavior, E114C mutant monomers accelerate the development of disease-modifying therapies. Their application supports translational research aimed at understanding and mitigating the molecular mechanisms underlying Parkinson’s disease and related neurodegenerative disorders. The alpha-synuclein (aSyn) E114C mutation facilitates a single site-specific conjugation with ATTO-488 maleimide that avoids any hindrance on fibrilization or cell entry that may be conferred by non-specific lysine targeting conjugations. This conjugation is ideal due to internal position relative to C-terminal truncation sites, proximity to the NAC, and lack of interference with recruitment in vitro or in primary neurons. Pre-formed fibrils (PFFs) generated with 5-25% fluorescently tagged E114C mutants have demonstrated a relative potency >80% compared to wild-type aSyn for inducing misfolding of endogenous aSyn, indicating no significant perturbation of seeding in living cells. ATTO-488 is a useful tool for identifying cell entry, as the addition of Trypan Blue to cultures prior to imaging will quench fluorescence of extracellular ATTO-488 conjugated aSyn. |
| References |
1.,Haney et al. 2016. Comparison of strategies for non-perturbing labeling of α-synuclein to study amyloidogenesis. Organic & Biomolecular Chemistry. DOI: 10.1039/c5ob02329g 2.,Karpowicz et al. 2017. Selective imaging of internalized proteopathic a-synuclein seeds in primary neurons reveals mechanistic insight into transmission of synucleinopathies. JBC. DOI: 10.1074/jbc.M117.780296 3.,Pieri et al. 2016. Structural and functional properties of prefibrillar α-synuclein oligomers. Scientific Reports. DOI: 10.1038/srep24526 |
Product Images
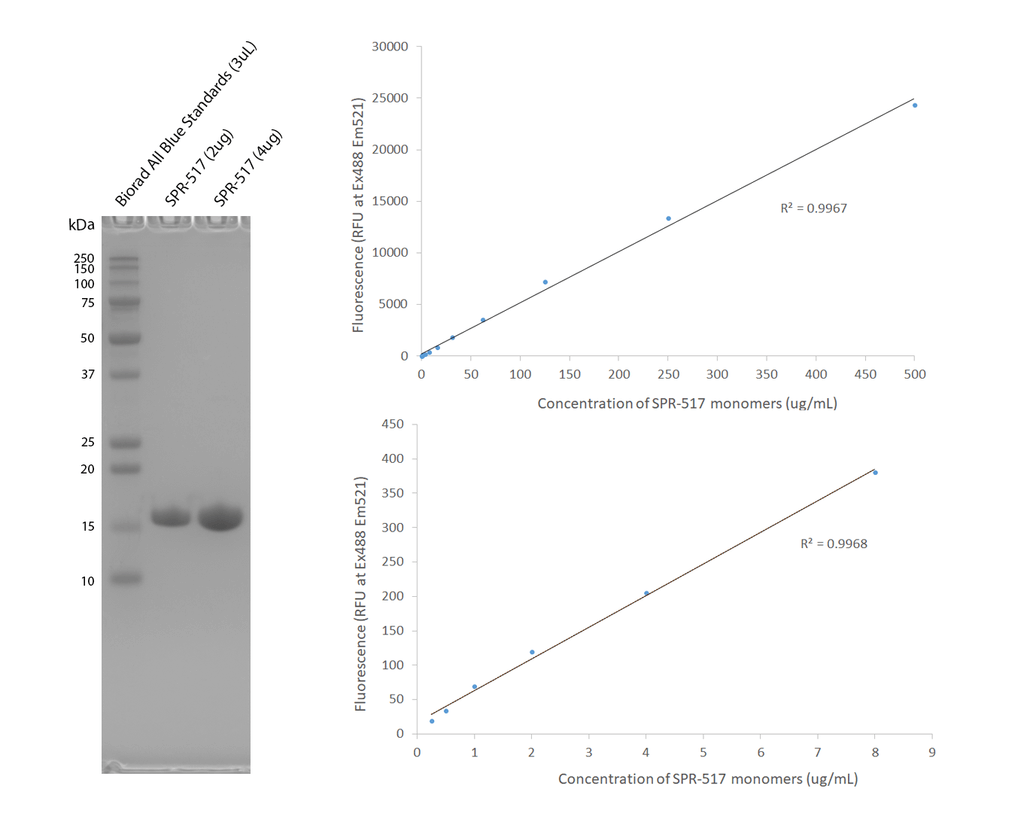
Purity and fluorescent signal of alpha-synuclein E114C-ATTO-488 monomers (SPR-517-A88). Left: SDS-PAGE analysis of SPR-517 monomers on a 12% Bis-Tris gel (left). Right: SPR-517 concentration and fluorescence (excitation 488nm, emission 521 nm) exhibit a linear relationship at all concentrations tested (250 ng/mL – 500 µg/mL).
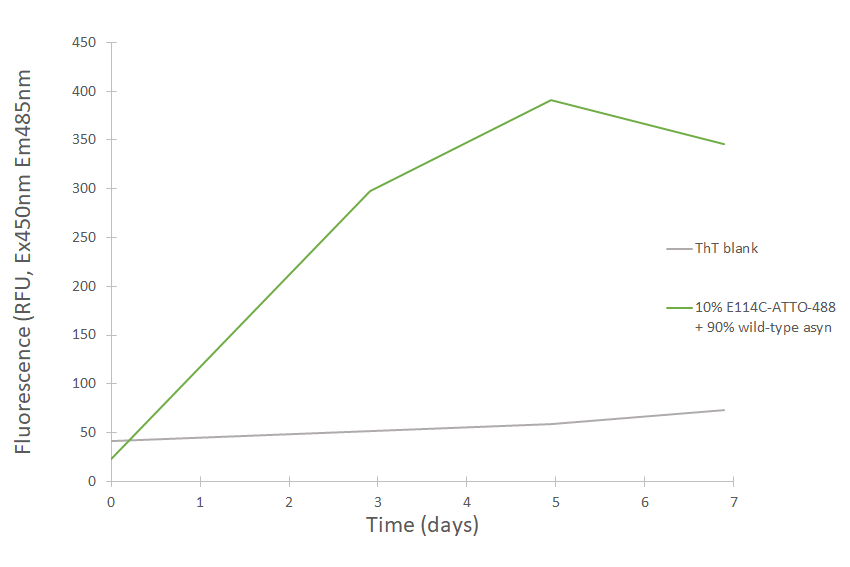
Fibril formation of ATTO-488 conjugated alpha-synuclein E114C monomers (SPR-517-A88). Fibrils were generated from a mixture of 10% E114C-ATTO-488 conjugated monomers and 90% wild-type monomers shaken 1000 rpm at 37oC for seven days in 1X PBS pH 7.4. Prior to measurement, a sample was taken, diluted to 0.5 mg/mL in 1X PBS pH 7.4 with 25 µM ThT and mixed. Excitation 450nm, emission 485 nm. Note: overall fibril ThT signal is dampened due to overlapping Atto-488 absorption maxima with ThT emission maxima.






















Reviews
There are no reviews yet.