Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein N-Terminal Acetylated Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-331) |
| Certificate of Analysis | Acetylation cofirmed via ESI mass spectrometry. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-332 |
Biological Description
| Alternative Names | N-acetylated alpha-synuclein, N-terminal acetylated alpha-synuclein, Alpha-synuclein, Alpha-synuclein aggregates, Alpha-synuclein seed, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, Parkinson's disease familial 1 Protein, N-terminal Ac-αsyn, Asyn |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Cellular Localization | Cytoplasm, Membrane, Nucleus |
| Accession Number | NP_000336.1 |
| Gene ID | 6622 |
| Swiss Prot | P37840 |
| Scientific Background |
Alpha-synuclein (α-syn), a neuronal protein encoded by the SNCA gene, plays a central role in synaptic function and plasticity. In its monomeric form, α-syn is subject to post-translational modifications, with N-terminal acetylation being one of the most prevalent and biologically significant. This modification alters the protein’s structural dynamics, membrane affinity, and aggregation behavior, making it a key factor in the onset and progression of neurodegenerative diseases. N-terminal acetylation enhances α-syn’s ability to adopt conformations that favor oligomerization and fibril formation—processes intimately linked to the development of synucleinopathies such as Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. Acetylated α-syn monomers exhibit increased interactions with lipid membranes, influencing vesicle trafficking and potentially disrupting neuronal homeostasis. Studies have demonstrated that acetylated α-syn monomers possess distinct biophysical properties compared to their non-acetylated counterparts, including altered aggregation kinetics and toxicity profiles. These differences suggest that N-terminal acetylation may serve as a molecular switch that modulates α-syn’s transition from a functional monomer to a pathogenic aggregate. Understanding the role of N-terminal acetylated α-syn monomers is critical for identifying early biomarkers and developing targeted therapies. By focusing on this specific molecular species, researchers aim to design interventions that prevent aggregation, enhance clearance, or stabilize native conformations, offering promising strategies to combat neurodegenerative diseases at their molecular roots. |
| References |
1. “Genetics Home Reference: SNCA”. US National Library of Medicine. (2013). 2. Zhang L., et al. (2008) Brain Res. 1244: 40-52. 3. Alim M.A., et al. (2002) J Biol Chem. 277(3): 2112-2117. 4. Kokhan V.S., Afanasyeva M.A., Van'kin G. (2012) Behav. Brain. Res. 231(1): 226-230. 5. Spillantini M.G., et al. (1997) Nature. 388(6645): 839-840. 6. Mezey E., et al. (1998) Nat Med. 4(7): 755-757. 7. Anderson, J.P. et al. (2006) J Biol Chem. 2006;281:29739–29752. 8. Vinueza-Gavilanes, R. et al. (2020) Neurobiol Dis. 137, 2020, Article 104783 |
Product Images
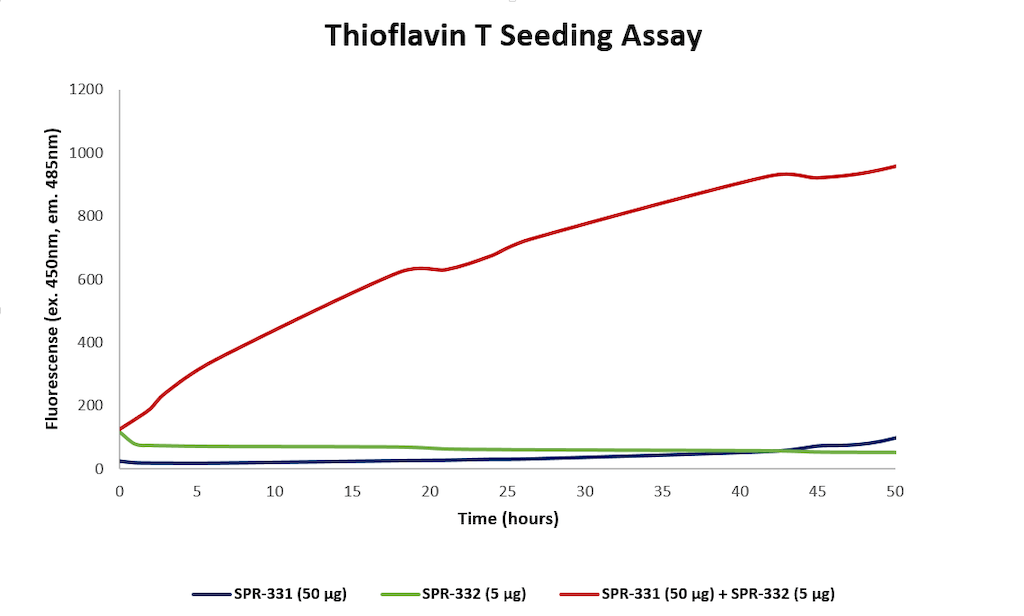
Thioflavin T assay for N-terminal acetylated alpha synuclein monomers (SPR-331) and pre-formed fibrils (PFFs) (SPR-332). Increased fluorescence, indicative of beta-sheet formation, was seen when 50 ug N-acetylated monomer and 5 ug N-acetylated fibril were combined, compared to monomer or fibril alone.






















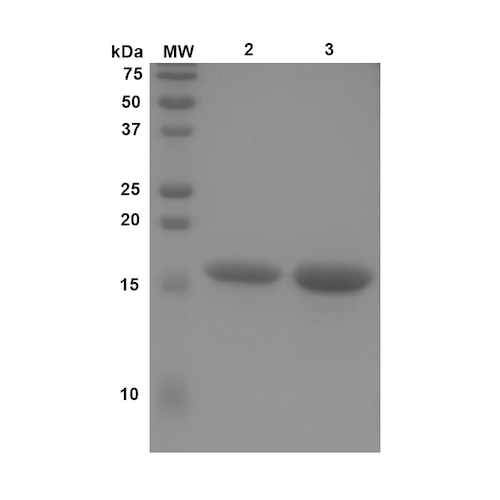
Reviews
There are no reviews yet.