| Product Name | Alpha Synuclein N-Terminal Acetylated Pre-formed Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
Human Recombinant N-Terminal Acetylated Alpha Synuclein PFFs (Type 1) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS-PAGE, In vivo assay, In vitro assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | 2 mg/mL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | Full Length | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | ~14.46kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Activity | Seeds aggregation of N-acetylated alpha synuclein monomer (SPR-331) in thioflavin T assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein N-Terminal Acetylated Pre-formed Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-332) |
| Certificate of Analysis | Acetylation cofirmed via ESI mass spectrometry. |
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-331. |
Biological Description
| Alternative Names | N-acetylated alpha-synuclein, N-terminal acetylated alpha-synuclein, Alpha-synuclein, Alpha-synuclein aggregates, Alpha-synuclein seed, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, Parkinson's disease familial 1 Protein, N-terminal Ac-αsyn, Asyn, Alpha Synuclein PFFs |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Cellular Localization | Cytoplasm, Membrane, Nucleus |
| Accession Number | NP_000336.1 |
| Gene ID | 6622 |
| Swiss Prot | P37840 |
| Scientific Background |
Alpha-synuclein (α-syn), a neuronal protein encoded by the SNCA gene, is central to synaptic function and implicated in the pathogenesis of synucleinopathies such as Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. Post-translational modifications, particularly N-terminal acetylation, significantly influence α-syn’s structural conformation, membrane interactions, and aggregation behavior. Pre-formed fibrils (PFFs) generated from N-terminally acetylated α-synuclein exhibit enhanced stability and seeding capacity compared to non-acetylated counterparts. These acetylated PFFs more effectively recruit endogenous α-syn monomers, accelerating the formation of toxic oligomers and fibrillar aggregates. This prion-like propagation of misfolded α-syn disrupts synaptic integrity, impairs mitochondrial function, and activates neuroinflammatory pathways, contributing to progressive neuronal degeneration. Experimental models using N-terminally acetylated α-syn PFFs demonstrate robust induction of Lewy body-like inclusions, synaptic dysfunction, and motor deficits, closely recapitulating human disease phenotypes. These models are instrumental in dissecting early pathogenic events and identifying molecular targets for therapeutic intervention. The study of N-terminal acetylated α-syn PFFs provides critical insights into the molecular mechanisms driving synucleinopathies. By understanding how acetylation modulates fibril formation and toxicity, researchers can develop targeted strategies to inhibit aggregation, block intercellular transmission, and enhance clearance of pathological species. These efforts are essential for advancing disease-modifying therapies and improving outcomes for individuals affected by neurodegenerative disorders. |
| References |
1. “Genetics Home Reference: SNCA”. US National Library of Medicine. (2013). 2. Zhang L., et al. (2008) Brain Res. 1244: 40-52. 3. Alim M.A., et al. (2002) J Biol Chem. 277(3): 2112-2117. 4. Kokhan V.S., Afanasyeva M.A., Van'kin G. (2012) Behav. Brain. Res. 231(1): 226-230. 5. Spillantini M.G., et al. (1997) Nature. 388(6645): 839-840. 6. Mezey E., et al. (1998) Nat Med. 4(7): 755-757. 7. Anderson, J.P. et al. (2006) J Biol Chem. 2006;281:29739–29752. 8. Vinueza-Gavilanes, R. et al. (2020) Neurobiol Dis. 137, 2020, Article 104783 |
Product Images
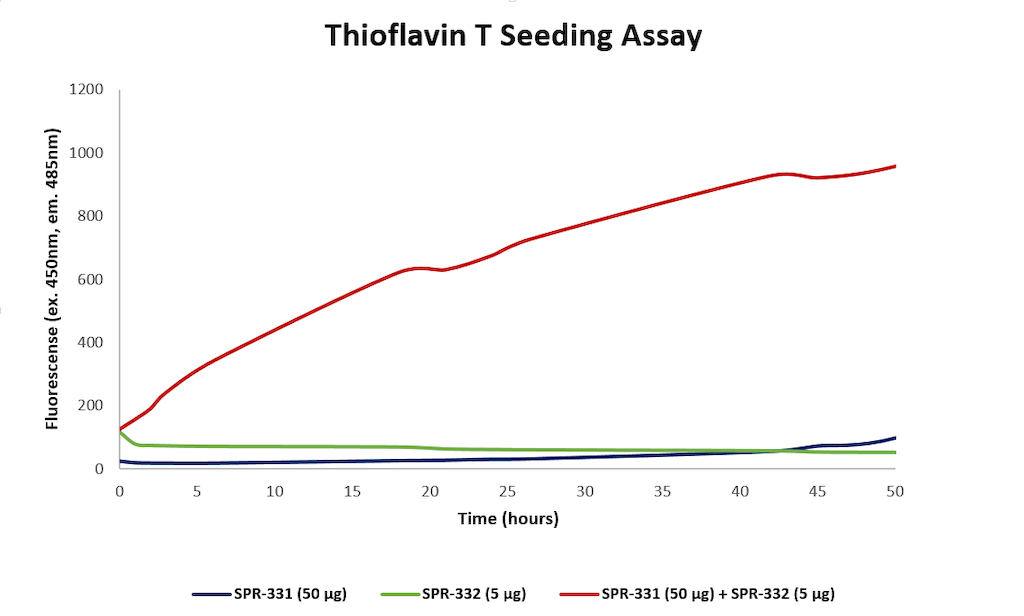
Thioflavin T assay for N-terminal acetylated alpha synuclein monomers (SPR-331) and pre-formed fibrils (PFFs) (SPR-332). Increased fluorescence, indicative of beta-sheet formation, was seen when 50 ug N-acetylated monomer and 5 ug N-acetylated fibril were combined, compared to monomer or fibril alone.























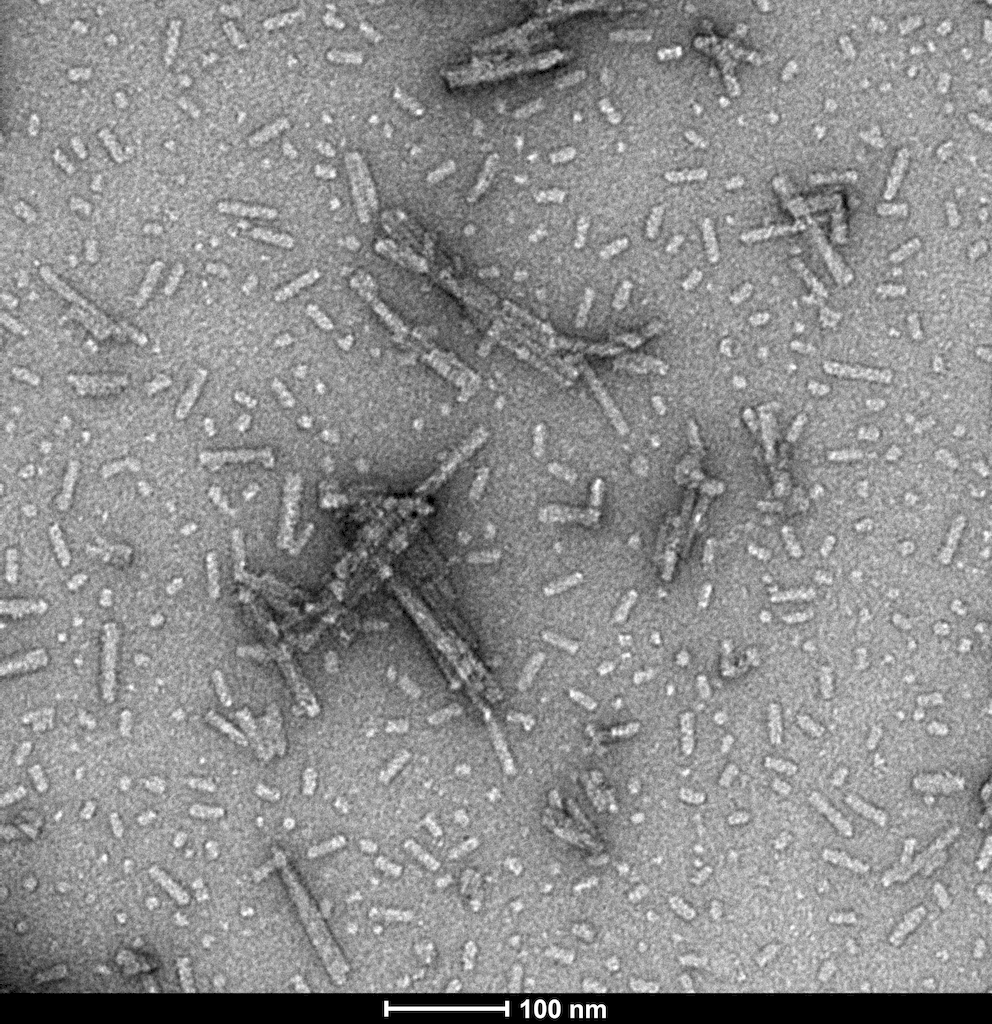

StressMarq Biosciences :
Based on validation through cited publications.