Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein Filaments (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-450) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-321. |
Biological Description
| Alternative Names | Alpha-synuclein filaments, Alpha-synuclein aggregates, Alpha-synuclein PFF, Alpha-synuclein PFFs, Alpha-synuclein seed, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, Parkinson's disease familial 1 Protein, Alpha-synuclein protofibrils, Alpha-synuclein proto-fibrils, Asyn |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Cellular Localization | Cytoplasm, Membrane, Nucleus |
| Accession Number | NP_000336.1 |
| Gene ID | 6622 |
| Swiss Prot | P37840 |
| Scientific Background |
Alpha-synuclein filaments are hallmark pathological structures in neurodegenerative diseases such as Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. These insoluble fibrillar aggregates arise from the misfolding and self-assembly of alpha-synuclein, a presynaptic neuronal protein encoded by the SNCA gene. Under pathological conditions, native alpha-synuclein undergoes conformational changes that promote β-sheet-rich filament formation. These filaments accumulate in neuronal and glial cells, disrupting essential cellular functions including proteostasis, mitochondrial dynamics, and synaptic signaling. Their presence correlates with progressive neurodegeneration and clinical decline. Recent advances in cryo-electron microscopy have revealed distinct filament structures associated with different synucleinopathies, suggesting disease-specific conformers with prion-like properties. These filaments can seed the misfolding of native alpha-synuclein, facilitating the spread of pathology across brain regions. Alpha-synuclein filaments are also being explored as biomarkers for early diagnosis and disease staging. Their unique biochemical signatures and spatial distribution offer promising avenues for targeted imaging and therapeutic intervention. Strategies aimed at inhibiting filament formation, promoting clearance, or stabilizing native alpha-synuclein are under active investigation. In summary, alpha-synuclein filaments are not only structural indicators of disease but also active drivers of neurodegeneration. Understanding their formation, propagation, and cellular impact is essential for developing precision therapies and improving outcomes for patients with synucleinopathies. |
| References |
1. “Genetics Home Reference: SNCA”. US National Library of Medicine. (2013). 2. Zhang L., et al. (2008) Brain Res. 1244: 40-52. 3. Alim M.A., et al. (2002) J Biol Chem. 277(3): 2112-2117. 4. Kokhan V.S., Afanasyeva M.A., Van'kin G. (2012) Behav. Brain. Res. 231(1): 226-230. 5. Spillantini M.G., et al. (1997) Nature. 388(6645): 839-840. 6. Mezey E., et al. (1998) Nat Med. 4(7): 755-757. |






















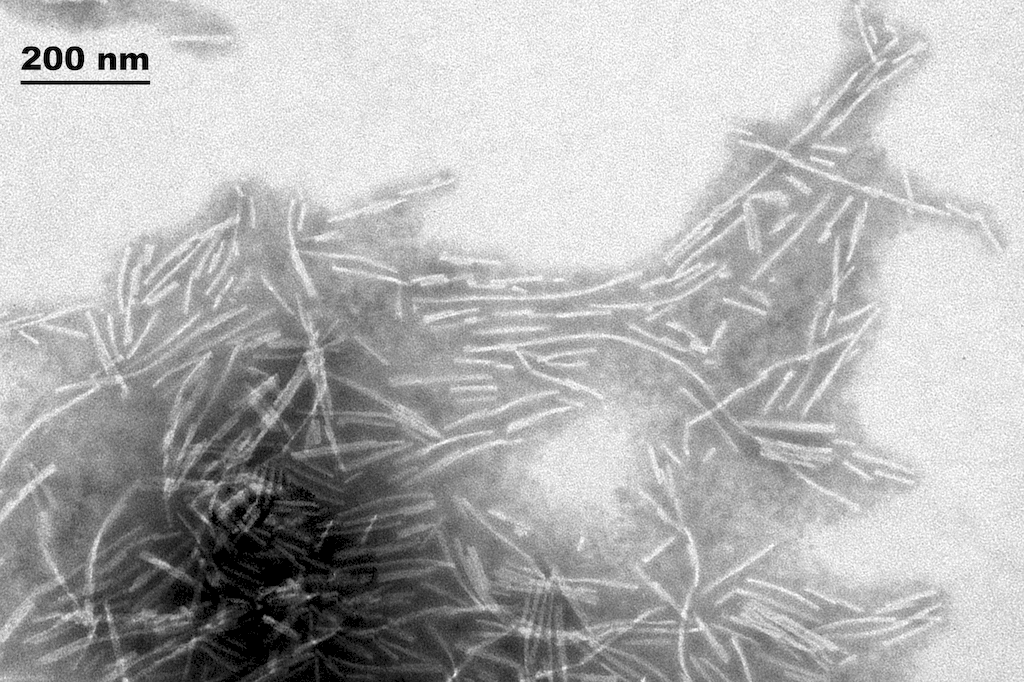
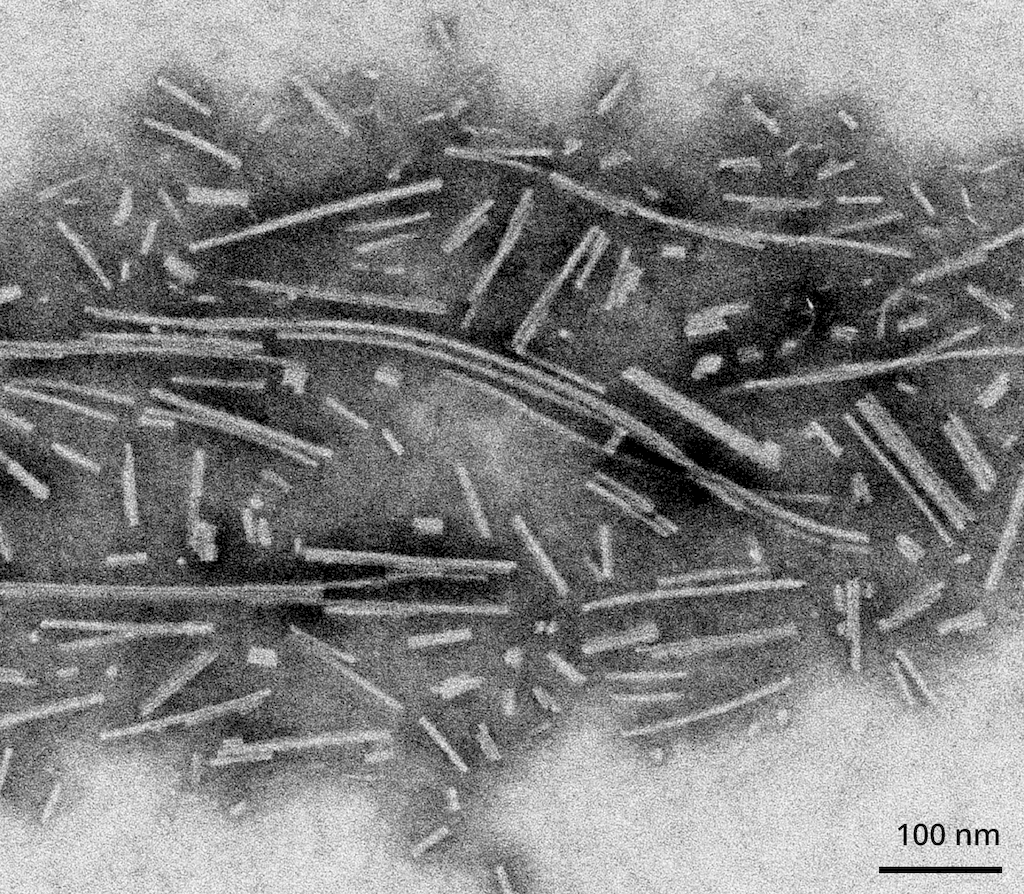
Reviews
There are no reviews yet.