Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Rat Recombinant Alpha Synuclein Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-481) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-482 |
Biological Description
| Alternative Names | Alpha-synuclein, Alpha synuclein, Asyn, SNCA, NACP, PARK1, PARK4, PD1, Synuclein alpha, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, Synuclein Alpha-140, SYN, Parkinson's disease familial 1 Protein Protein |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Cellular Localization | Cytoplasm, Membrane, Nucleus |
| Accession Number | NP_062042.1 |
| Gene ID | 29219 |
| Swiss Prot | P37377 |
| Scientific Background |
Alpha-synuclein is a neuronal protein that plays a central role in synaptic vesicle trafficking, neurotransmitter release, and dopamine regulation. In its native monomeric form, alpha-synuclein contributes to normal neuronal function by enhancing vesicle priming and fusion, and by acting as a molecular chaperone for SNARE complex assembly. In neurodegenerative diseases such as Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy, alpha-synuclein undergoes pathological misfolding and aggregation. While oligomers and fibrils are widely studied for their neurotoxicity, monomeric alpha-synuclein is increasingly recognized as a critical starting point for understanding disease initiation. Subtle conformational changes in monomers—triggered by mutations, post-translational modifications, or environmental stress—can prime the protein for aggregation and disrupt its physiological roles. Alpha-synuclein monomers are essential tools in mechanistic studies aimed at identifying early biomarkers and therapeutic targets. Their use in biochemical and cellular models enables high-resolution analysis of folding dynamics, membrane interactions, and protein-protein associations. These insights inform strategies to stabilize native alpha-synuclein and prevent its pathological transformation. |
| References |
1. “Genetics Home Reference: SNCA”. US National Library of Medicine. (2013). 2. Zhang L., et al. (2008) Brain Res. 1244: 40-52. 3. Alim M.A., et al. (2002) J Biol Chem. 277(3): 2112-2117. 4. Kokhan V.S., Afanasyeva M.A., Van'kin G. (2012) Behav. Brain. Res. 231(1): 226-230. 5. Spillantini M.G., et al. (1997) Nature. 388(6645): 839-840. 6. Mezey E., et al. (1998) Nat Med. 4(7): 755-757. 7. Polymeropoulos, M. H. (1998). Science. 276(5321), 2045–2047 8. Conway, K.E., et al. (1998). Nat Med. 4(11):1318-20 |
Product Images
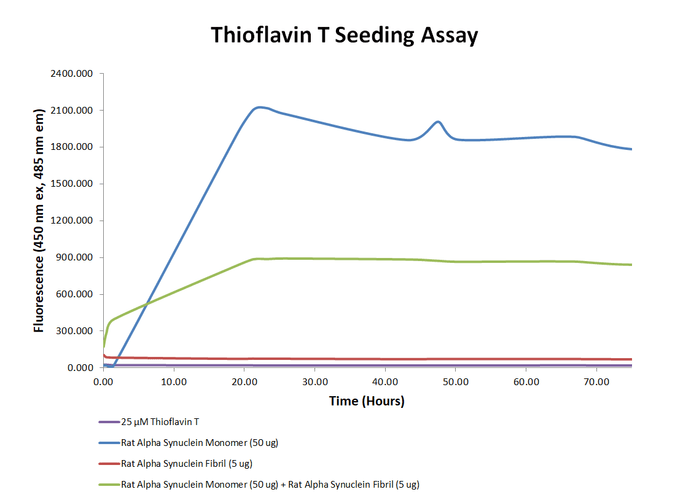
Thioflavin T is a fluorescent dye that binds to beta sheet-rich structures, such as those in alpha synuclein monomers and fibrils. Upon binding, the emission spectrum of the dye experiences a red-shift, and increased fluorescence intensity. The Rat Alpha Synuclein monomer (SPR-481) is very active and was able to form more beta-sheet structure alone than with the combination of monomer and fibril. In combination, the fibril (SPR-482) is the majority of the seed.

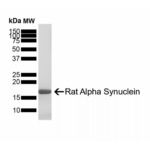





















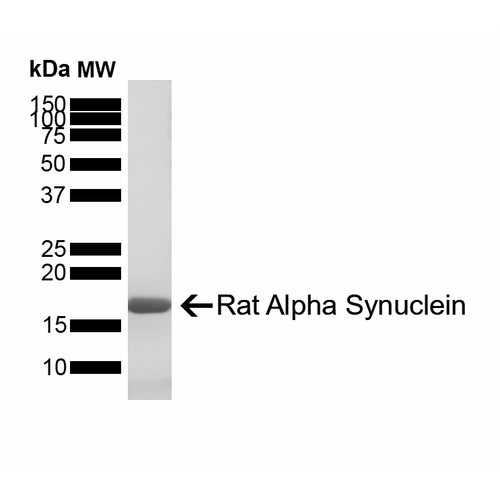
Reviews
There are no reviews yet.