| Product Name | Alpha Synuclein pSer129 Pre-Formed Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
Human Recombinant Alpha Synuclein pSer129 PFFs (phosphorylated at position 129) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS-PAGE, In vivo assay, In vitro assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | 2 mg/ml or 5 mg/ml | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amino Acid Sequence | MDVFMKGLSK AKEGVVAAAE KTKQGVAEAA GKTKEGVLYV GSKTKEGVVH GVATVAEKTK EQVTNVGGAV VTGVTAVAQK TVEGAGSIAA ATGFVKKDQL GKNEEGAPQE GILEDMPVDP DNEAYEMPSE EGYQDYEPEA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
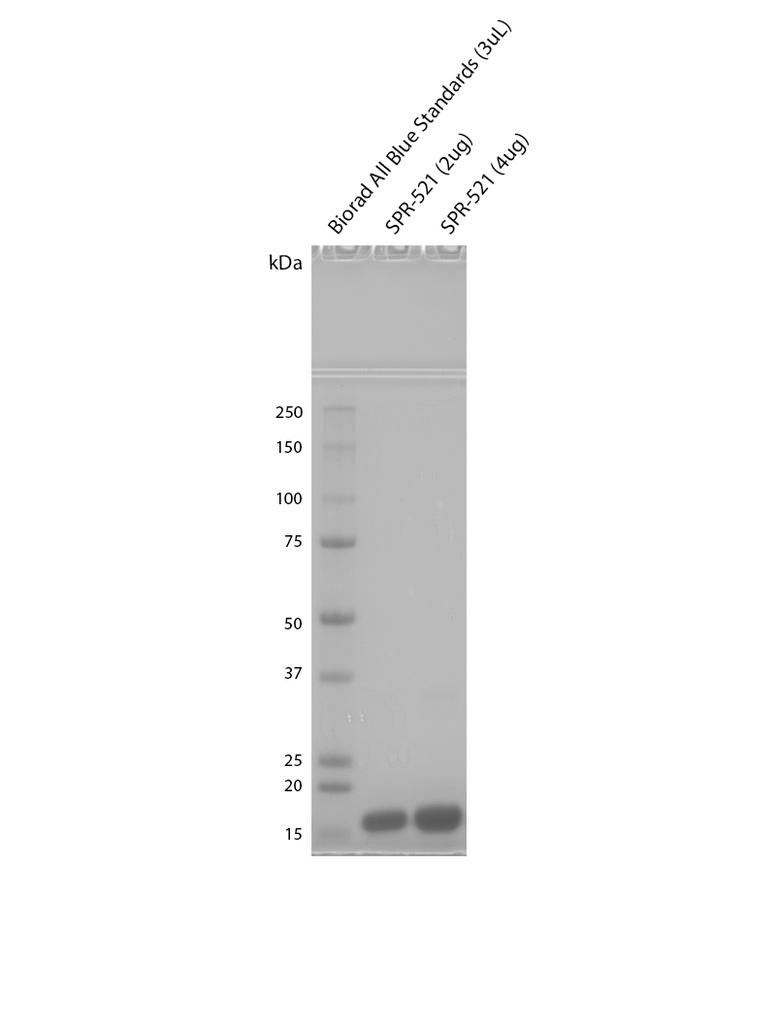
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | 140 AA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | ~14.46 kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein pSer129 Pre-Formed Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-521) |
| Certificate of Analysis | Protein certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. For corresponding monomers, see Catalog# SPR-520. The unphosphorylated construct is Catalog# SPR-322. |
Biological Description
| Alternative Names | Alpha-synuclein, Alpha synuclein, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, PD1, PARK4, Synuclein Alpha, Alpha Synuclein pSer129, Asyn, Alpha Synuclein PFFs |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Swiss Prot | P37840 |
| Scientific Background |
Alpha-synuclein (α-synuclein), a neuronal protein encoded by the SNCA gene, plays a central role in synaptic vesicle trafficking and neurotransmitter release. In neurodegenerative diseases such as Parkinson’s disease, α-synuclein aggregates into fibrillar structures, with phosphorylation at serine 129 (pSer129) being the most prevalent post-translational modification observed in pathological inclusions like Lewy bodies. Pre-formed fibrils (PFFs) composed of α-synuclein phosphorylated at Ser129 replicate key features of disease-associated aggregates. These pSer129 PFFs exhibit enhanced seeding capacity and altered structural properties, making them highly effective in inducing endogenous α-synuclein aggregation in cellular and animal models. Their ability to mimic disease-relevant pathology—including fibril morphology, neurotoxicity, and prion-like propagation—makes them indispensable tools for mechanistic studies. In neurodegenerative disease research, pSer129 PFFs enable detailed investigation into the role of phosphorylation in modulating α-synuclein aggregation, intercellular transmission, and neuroinflammatory responses. They are also used to evaluate therapeutic strategies targeting phosphorylation-dependent aggregation pathways, promoting clearance of toxic species, and restoring neuronal function. By modeling the biochemical and pathological characteristics of α-synuclein aggregates, pSer129 PFFs accelerate translational research and drug development for Parkinson’s disease and related synucleinopathies. StressMarq's Alpha Synuclein Ser129 Pre-Formed Fibrils are generated in-vitro from purified phosphorylated monomer and phosphorylation is confirmed with our anti-ASYN pS129 monoclonal antibody (catalog# SMC-600). |
| References |
1. Okochi et al. 2000. Constitutive phosphorylation of the Parkinson’s Disease Associated a-Synuclein. The Journal of Biological Chemistry. DOI: 10.1074/jbc.275.1.390 2. Fujiwara et al. 2002. α-Synuclein is phosphorylated in synucleinopathy lesions. Nature Cell Biology. DOI: 10.1038/ncb748 3. Magalhaes and Lashuel. 2002. Opportunities and challenges of alpha-synuclein as a potential biomarker for Parkinson’s disease and other synucleinopathies. Npj Parkinsons Disease. DOI: 10.1038/s41531-022-00357-0 |
Product Images
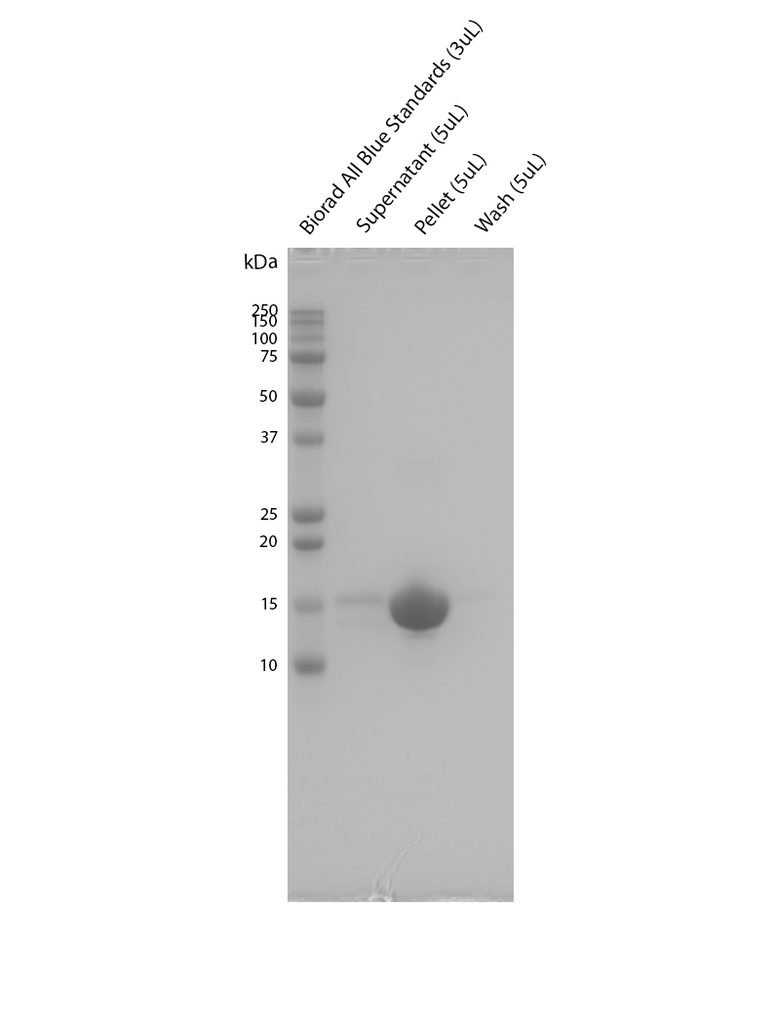
Sedimentation assay on Human Recombinant Alpha Synuclein pSer129 Pre-Formed Fibrils (SPR-521). Samples were spun down at 15,000 x g, washed, and then spun down again. Fibril samples are prepared in denaturing conditions prior to running on the gel. SDS-PAGE analysis on a 12% Bis-Tris gel shows that the majority of the fibril is insoluble.
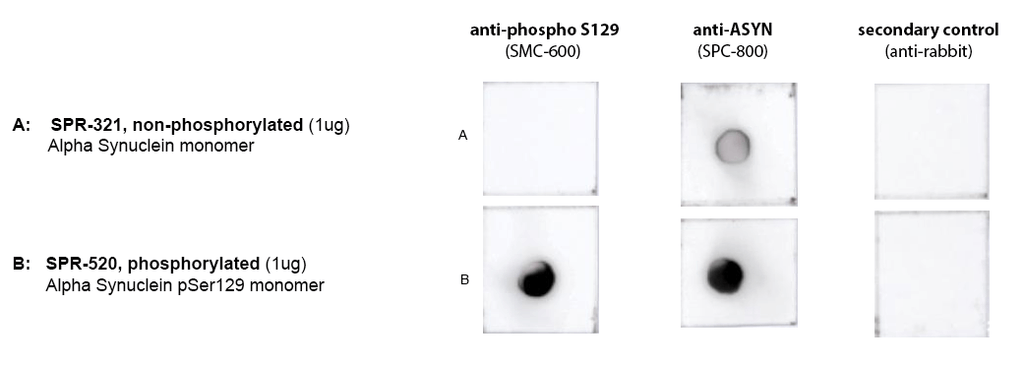
Dot Blot of purified Human Recombinant Alpha Synuclein pSer129 Monomers (starting material for SPR-521) using Stressmarq’s SMC-600 (anti-ASYN pS129) and SPC-800 (anti-ASYN) confirming phosphorylation in SPR-520, compared to SPR-321. Protein was blotted on nitrocellulose, incubated with 1:1000 primary antibodies and/or 1:4000 secondary antibodies. Secondary control is goat-anti rabbit:HRP. Exposed 1 second.
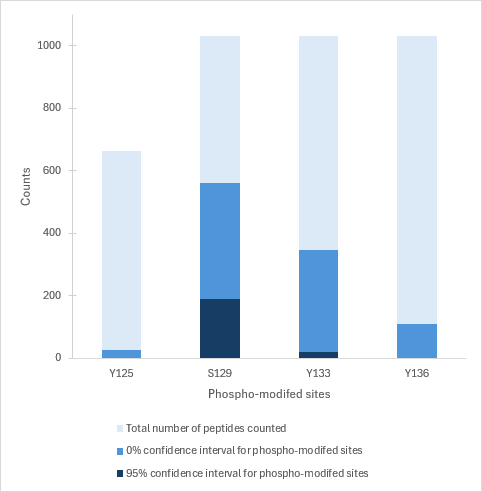
Modified/total phosphorylation PTM spectrum counts of 4 sites on human alpha synuclein pSer129 monomers (starting material for SPR-521) as determined by mass spectrometry (ScaffoldPTM, localization probability = 100% at both 0% and 95% min. localization). CNBr digestion was used to accurately determine the presence of phosphorylation at and around S129. Note that total counts may include longer peptides where phosphorylation may be more difficult to detect, and most phospho-modified sites that were detected are at S129.
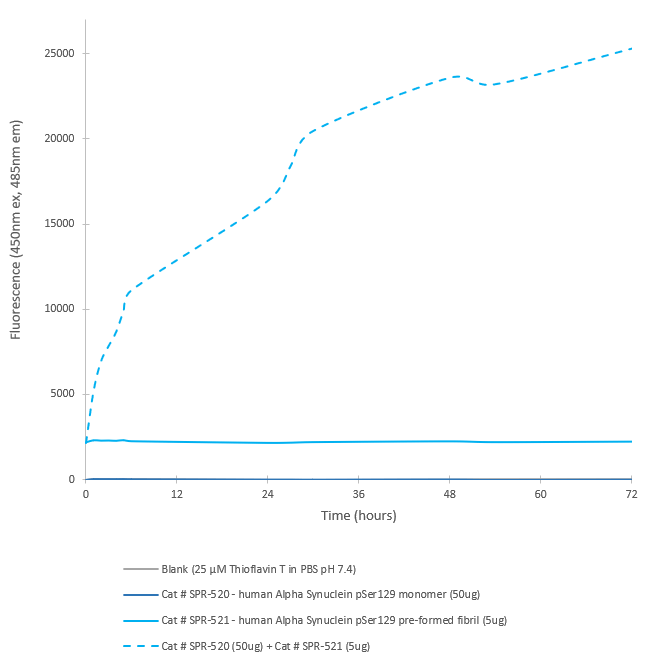
In vitro seeding activity of Human Recombinant Alpha Synuclein pSer129 Pre-Formed Fibrils (SPR-521) in ThT assay. Alpha Synuclein pSer129 Pre-Formed Fibrils seed fibril formation of Alpha Synuclein pSer129 (SPR-520) over 72 hours. Reactions (100uL) shaken at 600 rpm in Greiner-Bio 96 Well Non-Binding Cell Culture Microplates, Black (Greiner-Bio Catalog #655900) at 37oC in the presence of 25 uM ThT and read with an XPS Microplate Reader set at 450nmex/485nmem.

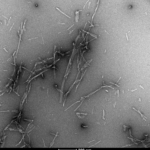
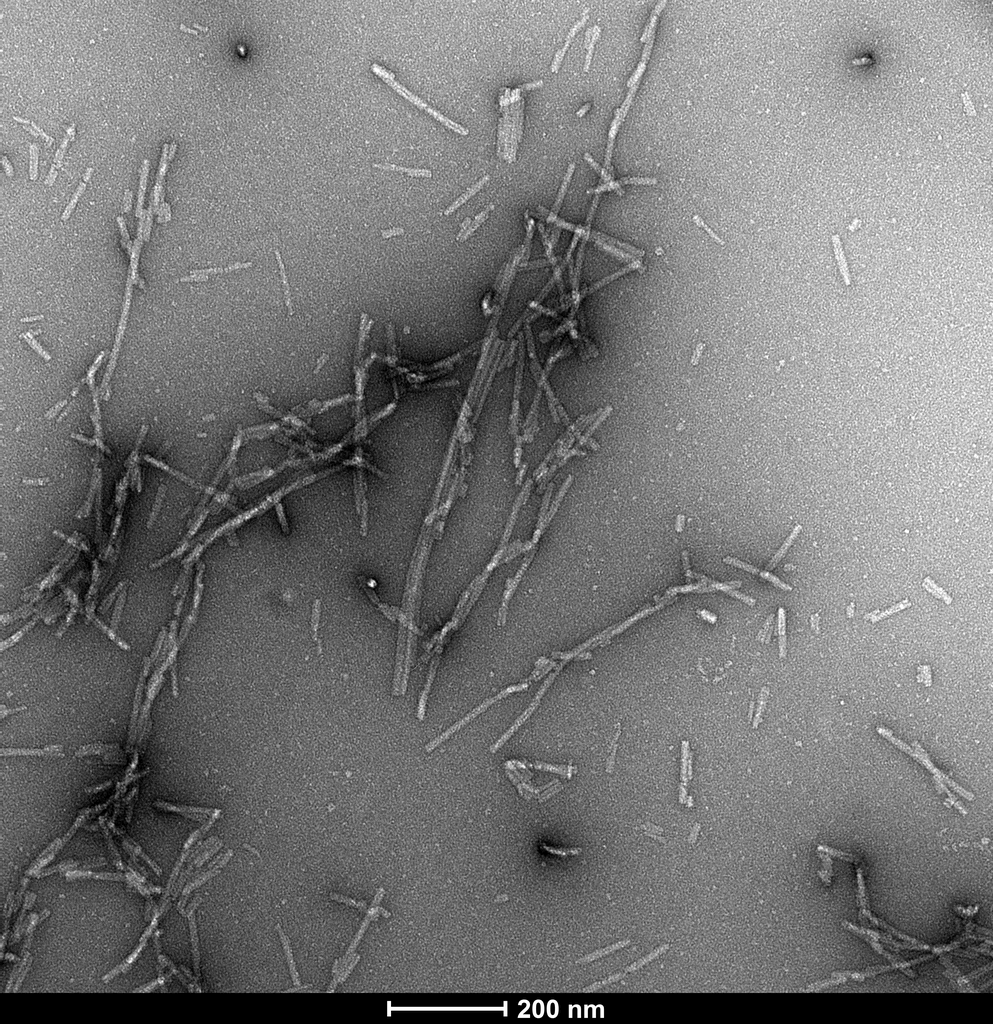
Atomic force microscopy analysis of Alpha Synuclein pSer129 Pre-Formed Fibrils (SPR-521) diluted to 0.02 mg/mL with dH2O mounted on freshly cleaved mica, washed, dried and analyzed with tapping mode. Representative images are 5 x 5 µm x-y (left) and 2 x 2 µm x-y (right), both with a Z-range set at 17nm.






















Reviews
There are no reviews yet.