Properties
| Storage Buffer | 1X PBS pH7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein S129A Mutant Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-505) |
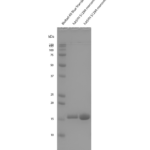
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-506 |
Biological Description
| Alternative Names | Alpha-synuclein, Alpha synuclein, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, NACP, SNCA, PARK1, SYN, PD1, PARK4, Synuclein Alpha, Parkinson's disease familial 1 Protein, Alpha Synuclein S129A, Asyn |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Swiss Prot | P37840-1 |
| Scientific Background |
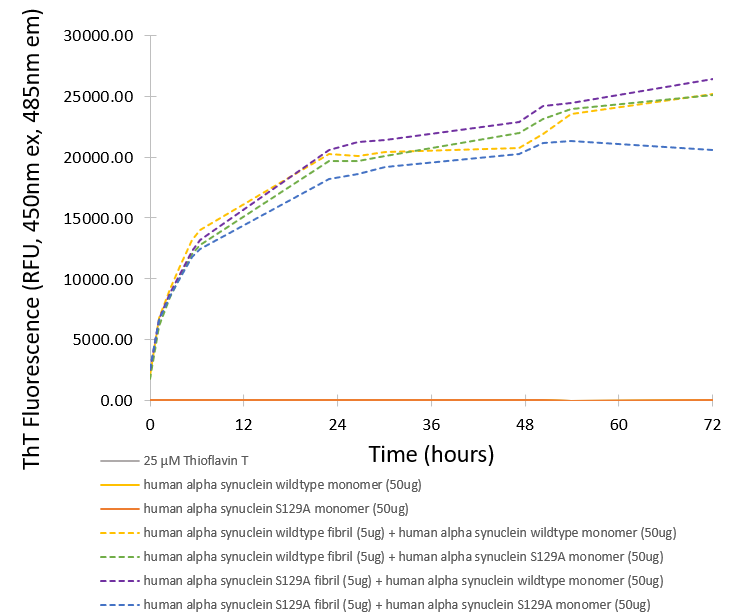
Alpha-synuclein (α-synuclein), encoded by the SNCA gene, is a presynaptic neuronal protein involved in synaptic vesicle trafficking, neurotransmitter release, and SNARE-complex assembly. Phosphorylation at serine 129 (S129) is a hallmark of pathological α-synuclein found in Lewy bodies, the defining feature of Parkinson’s disease and related synucleinopathies. The S129A mutation replaces serine with alanine, preventing phosphorylation at this critical site. This modification allows researchers to isolate the functional consequences of phosphorylation-independent α-synuclein behavior. S129A mutant monomers exhibit altered aggregation kinetics, membrane interactions, and cellular toxicity profiles, making them a valuable tool for dissecting the molecular mechanisms of α-synuclein pathology. In neurodegenerative disease models, S129A monomers help clarify the role of post-translational modifications in α-synuclein misfolding, oligomerization, and neurotoxicity. Their use enables targeted investigation into how phosphorylation influences protein conformation, intracellular trafficking, and interactions with cellular components such as mitochondria and lysosomes. By providing a phosphorylation-null background, Alpha-Synuclein S129A mutant monomers support the development of therapeutic strategies aimed at modulating α-synuclein structure and function. They are instrumental in screening compounds that prevent aggregation, enhance clearance, or restore synaptic integrity, thereby accelerating translational research in Parkinson’s disease and other neurodegenerative disorders. |
| References |
1. Xu, Deng and Qing. 2015. The phosphorylation of α-synuclein: development and implication for the mechanism and therapy of the Parkinson's disease. Journal of Neurochemistry. https://doi.org/10.1111/jnc.13234 2. Ramalingam et al. 2023. Dynamic physiological α-synuclein S129 phosphorylation is driven by neuronal activity. NPJ Parkinsons Dis. doi: 10.1038/s41531-023-00444-w. |






















Reviews
There are no reviews yet.