Properties
| Storage Buffer | 1X PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Alpha Synuclein S87N Mutant Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-499) |
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-500 |
Biological Description
| Alternative Names | Alpha Synuclein S87N, Alpha-synuclein, Alpha synuclein, SNCA, synuclein, NACP, Non-A beta component of AD amyloid, Non-A4 component of amyloid precursor, PARK1, SYN, Parkinson's disease familial 1 Protein, Asyn |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Accession Number | NP_000336.1 |
| Gene ID | 6622 |
| Swiss Prot | P37840-1 |
| Scientific Background |
Alpha-synuclein (α-synuclein), encoded by the SNCA gene, is a neuronal protein critically involved in synaptic vesicle trafficking, neurotransmitter release, and SNARE-complex assembly. The S87N mutation represents a single amino acid substitution within the non-amyloid-β component (NAC) region, a domain essential for α-synuclein aggregation and membrane interactions. In its monomeric form, wild-type α-synuclein contributes to synaptic homeostasis by modulating vesicle fusion and exocytotic pore dynamics. However, the S87N mutant alters these physiological functions, potentially disrupting calcium-dependent exocytosis and impairing SNARE-complex formation. These changes may accelerate synaptic dysfunction and neuronal vulnerability, key features of Parkinson’s disease and related synucleinopathies. Research utilizing S87N mutant monomers has revealed their distinct biophysical properties, including altered aggregation kinetics and membrane binding affinity. These characteristics make them valuable tools for modeling early-stage neurodegeneration, enabling precise investigation of pathogenic mechanisms such as protein misfolding, intracellular trafficking defects, and neuroinflammation. By mimicking disease-relevant molecular events, S87N mutant monomers serve as a robust platform for screening therapeutic candidates aimed at stabilizing α-synuclein structure, preventing toxic oligomer formation, and restoring synaptic function. Their use in cellular and animal models enhances translational research targeting the molecular underpinnings of Parkinson’s disease and other α-synuclein-driven disorders. Human alpha synuclein S87N mutant (HuS87N) has Ser87 mutated to the equivalent mouse residue Asn87, effectively making it a human-mouse chimeric protein. Despite sequence differences at only seven residues, or 5% of the total 140 amino acids, the aggregation rate of wild-type mouse α-syn (MsWT) is faster than wild-type human α-syn (HuWT) in vitro. In wild-type mouse models, MsWT fibrils are more efficient than HuWT fibrils at inducing pathology of endogenous mouse α-syn (1). A53T or S87N substitutions in human α-syn substantially accelerate fibrilization rates in vitro (2,3). Chimeric HuS87N fibrils show enhanced pathogenicity to wild-type mouse neurons, greater than HuWT, HuA53T, and MsWT fibrils (4). HuS87N fibrils can be used as a more human-like alternative to MsWT fibrils to induce equivalent or greater endogenous α-syn seeding and pathology in wild-type mice. |
| References |
1. Masuda-Suzukake et al. 2013. Prion-like Spreading of Pathological α-synuclein in Brain. Brain. https://doi.org/10.1093/brain/awt037 2. Kang, K. et al. 2011. The A53T Mutation is Key in Defining the Differences in the Aggregation Kinetics of Human and Mouse α-synuclein. JACS. https://doi.org/10.1021/ja203979j 3. Ohgita, T. et al. 2023. Intramolecular Interaction Kinetically Regulates Fibril Formation by Human and Mouse Alpha-Synuclein. Sci Rep https://doi.org/10.1038/s41598-023-38070-4 4. Luk, K., C. et al. 2016. Molecular and Biological Compatibility with Host Alpha-Synuclein Influences Fibril Pathogenicity. Cell Rep. https://doi.org/10.1016/j.celrep.2016.08.053 |
Product Images
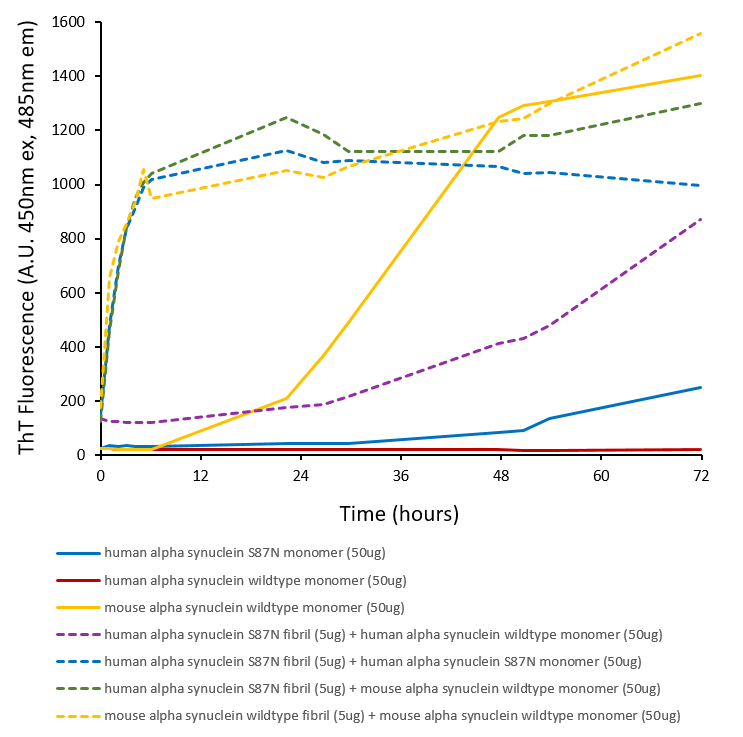
Fibril formation and seeding activity of human alpha synuclein S87N mutant measured by ThT in vitro. Human S87N mutant monomers self-aggregate faster than human wild-type monomers. Human S87N mutant pre-formed fibrils rapidly seed S87N mutant monomers and mouse wild-type monomers. Human S87N mutant fibrils also seed human wild-type monomers, although less aggressively than mouse wild-type monomers.






















Reviews
There are no reviews yet.