Properties
| Storage Buffer | Phosphate buffer (PB) pH 7.4 and 10 mM NaCl |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | N/A |
| Cite This Product | Human Synthetic (TFA preparation) Amyloid Beta 1-42 Oligomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-488) |
| Certificate of Analysis | Certified >95% pure using mass spec and HPLC. Low endotoxin <2.5 EU/mL @ 1mg/mL. |
Biological Description
| Alternative Names | Aβ 1-42, Amyloid beta, Beta amyloid peptide, Abeta, Abeta peptide, Amyloid beta precursor peptide, APP, Beta-APP42, Abeta42, A4, ABPP, APPI, Alzheimer disease amyloid A4, Alzheimer disease amyloid, Cerebral vascular amyloid peptide, PreA4, Protease nexin-II, PN-II |
| Research Areas | Alzheimer's Disease, Amyloid, Neurodegeneration, Neuroscience |
| Cellular Localization | Cell membrane, Intracellular Vesicles |
| Gene ID | 351 |
| Swiss Prot | P05067 |
| Scientific Background |
Amyloid beta (Aβ) peptides, particularly the 42-residue variant Aβ 1-42, are central to the pathogenesis of Alzheimer’s disease. While fibrillar aggregates form the hallmark amyloid plaques, it is the soluble oligomeric intermediates of Aβ 1-42 that are increasingly recognized as the most neurotoxic species. Aβ 1-42 oligomers disrupt neuronal function by impairing synaptic transmission, altering calcium homeostasis, and triggering oxidative stress and inflammation. These small aggregates interact with cell membranes, receptors, and intracellular signaling pathways, leading to progressive neurodegeneration. Their ability to seed further aggregation and propagate pathology across brain regions mirrors prion-like mechanisms observed in other proteinopathies. In research settings, Aβ 1-42 oligomers serve as a critical model for studying early-stage Alzheimer’s disease. They enable high-resolution analysis of misfolding kinetics, neurotoxicity, and cellular responses. Their relevance to human pathology makes them ideal for evaluating therapeutic strategies aimed at neutralizing oligomeric species, preventing aggregation, and restoring neuronal health. By capturing the transition from monomeric peptide to pathogenic aggregate, Aβ 1-42 oligomers provide a powerful platform for advancing biomarker discovery and the development of disease-modifying treatments in neurodegenerative research. StressMarq's Amyloid Beta 1-42 (Aβ42) Oligomers are generated from Amyloid Beta Peptide 1-42 pre-treated with 1,1,1,3,3,3-Hexafluoro-2-propanol (HFIP) as previously published (1,2). StressMarq's Aβ42 oligomers present as globular oligomers when observed under TEM and AFM, have a unique dimer/trimer and oligomer signal on a Western Blot with an anti-amyloid beta antibody, and exhibited dose‑dependent toxicity in primary rat cortical neurons. |
| References |
1. Stine et al. 2003. JBC. 278(13):11612-22. doi: 10.1074/jbc.M210207200 2. Ahmed et al. 2010. Nature Structural & Molecular Biology. 17(5):561-7. doi: 10.1038/nsmb.1799 3. Panza et al. 2019. Nat Rev Neurol. 15:73-88 https://doi.org/10.1038/s41582-018-0116-6 4. Shankar et al. 2008. Nat Med. 14(8):837-842. doi: 10.1038/nm1782 5. Chromy et al. 2003. Biochemistry. 42:12749-12760. doi: 10.1021/bi030029q 6. Kayed et al. 2003. Science. 300(5618): 486-489. doi: 10.1126/science.1079469 7. Want et al. 2016. JAMA Neurol. 73(9):1070-7. doi: 10.1001/jamaneurol.2016.2078 8. Kotzbauer et al. 2012. Arch Neurol. 69(10): 1326-1331. doi: 10.1001/archneurol.2012.1608 |
Product Images

AFM of amyloid beta 1-42 monomers (SPR-485, left), oligomers (SPR-488, middle) and fibrils (SPR-487, right). Atomic force microscopy analysis of 1.0 mg/mL samples diluted to 0.1 mg/mL in dH2O, mounted on freshly cleaved mica, washed, dried and analyzed with tapping mode. Representative images are 2.5 x 2.5 µm x-y with a z-range of 10 nm.
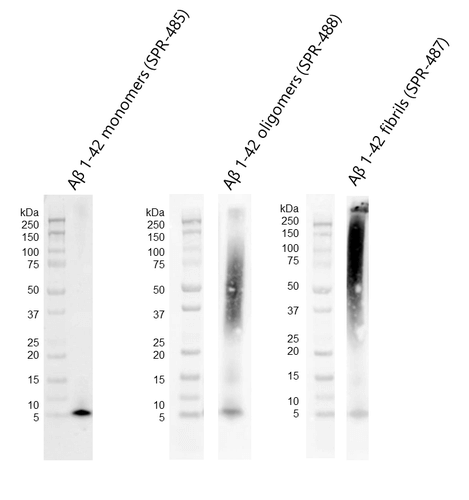
Western blot of amyloid beta 1-42 monomers (SPR-485, left), oligomers (SPR-488, middle) and fibrils (SPR-487, right) using anti-amyloid beta 6E10 antibody. Amyloid beta constructs at 160 pmol were run on 4-12% Bis-Tris SDS-PAGE, transferred to nitrocellulose in the presence of 0.02% v/v Tween-20, and blotted with 1:1000 mouse 6E10 primary antibody (Biolegend). Oligomers observed under TEM/AFM appear as distinct dimer/trimer bands at ~37-75 kDa on Western Blot with 6E10 antibody (middle). Fibrils observed under TEM/AFM appear as a distinct signal at greater than 100 kDa in the stacking gel (right).
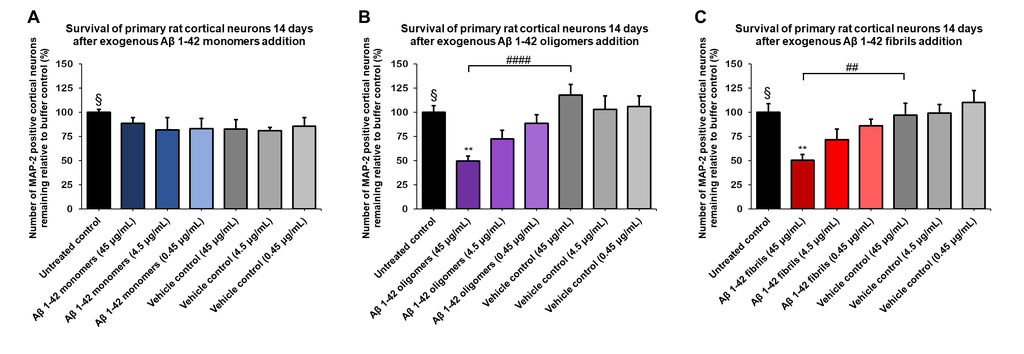
Amyloid beta 1-42 oligomers (SPR-488) and fibrils (SPR-487) show a dose-dependent toxicity to primary rat cortical neurons, but not monomers (SPR-485). Survival of rat primary cortical neurons 14 days after treatment with different concentrations of (A) monomers, (B) oligomers or (C) fibrils quantified by MAP2 positive neurons and expressed as a percentage of control. Fibrils and respective vehicle controls were initially sonicated in a Bioruptor. Test conditions were run in the same plate as untreated control and vehicle controls, which consisted of buffer without amyloid beta 1-42 protein. Data expressed as mean +/- s.e.m. (n=6). A global analysis of the data was performed using a one-way ANOVA followed by Dunnett’s test; ** p<0.01 stats vs control; ## p<0.01, #### p<0.0001 stats vs vehicle control. § represents untreated control condition.


























Reviews
There are no reviews yet.