Properties
| Storage Buffer | 10mM Hepes pH 7.4, 100mM NaCl |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau-381 (1N3R) Wild-Type Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-513) |
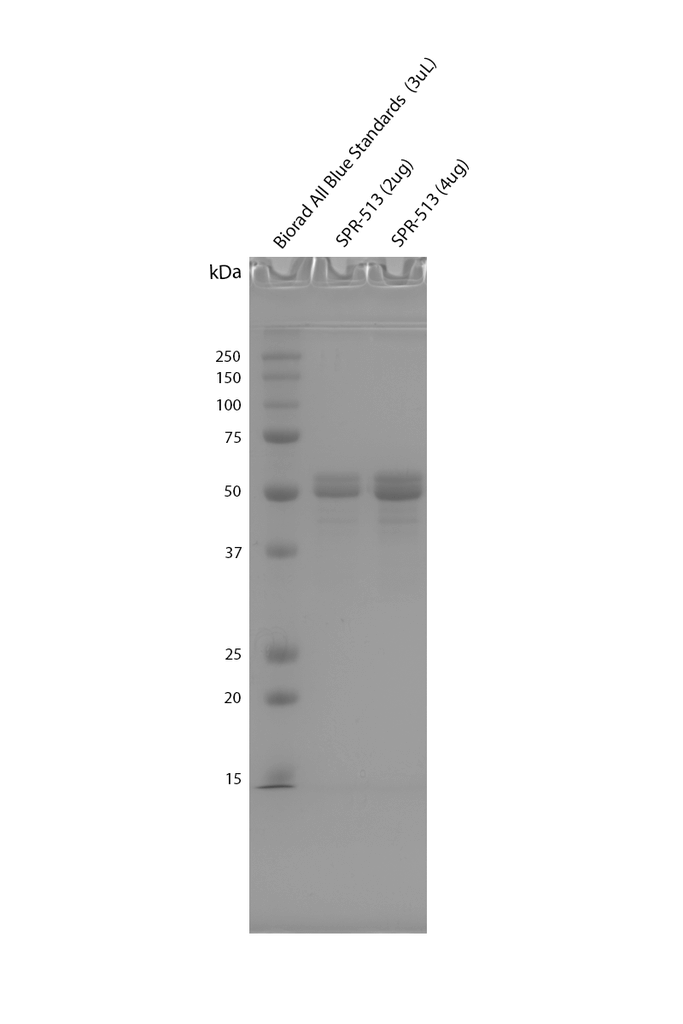
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & > 80% Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-514. |
Biological Description
| Alternative Names | Tau 381, Tau-B, Tau-D, Tau-40, MAPT, TAU, MTBT1, MTBT2, MAPTL, PPND, PPP1R103, FTDP-17, PHF-Tau, Paired Helical Filament-Tau, Neurofibrillary Tangle, Microtubule-associated tau, Isoform 3, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1 |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Tangles & Tau |
| Swiss Prot | P10636-4 |
| Scientific Background |
Tau-381, also known as the 1N3R isoform of the microtubule-associated protein tau (MAPT), is a naturally occurring variant composed of 381 amino acids. It includes one N-terminal insert and three microtubule-binding repeat domains, making it one of the shorter isoforms expressed in the adult human brain. This structural configuration contributes to its dynamic role in microtubule stabilization and neuronal plasticity. In healthy neurons, Tau-381 supports cytoskeletal organization and axonal transport. However, under pathological conditions, Tau-381 monomers can misfold and initiate aggregation into toxic oligomers and fibrils—key events in the development of tauopathies such as Alzheimer’s disease and Pick’s disease. The 3R isoforms, including Tau-381, are particularly relevant in diseases characterized by selective isoform expression and aggregation. Tau-381 wild-type monomers are widely used in neurodegenerative disease research to model early tau aggregation and misfolding. Their defined structure and aggregation behavior make them ideal for in vitro studies, structural analysis, and therapeutic screening. Researchers utilize these monomers to investigate the molecular mechanisms of tau toxicity, post-translational modifications, and interactions with cellular components. By enabling precise analysis of tau dynamics, Tau-381 monomers provide a robust platform for developing targeted therapies aimed at preventing aggregation, enhancing clearance, and restoring neuronal function. Their application accelerates translational research focused on mitigating tau-driven neurodegeneration. |
| References |
1. Goedert et al. 1989. Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer’s disease. Neuron. DOI: 10.1016/0896-6273(89)90210-9 2. Goedert, Eisenberg and Crowther. 2017. Propagation of Tau Aggregates and Neurodegeneration. Annual Review of Neuroscience. DOI: 10.1146/annurev-neuro-072116-031153 3. Dregni et al. 2022. Fluent molecular mixing of Tau isoforms in Alzheimer’s disease neurofibrillary tangles. Nature communications. DOI: 10.1038/s41467-022-30585-0 |
Product Images
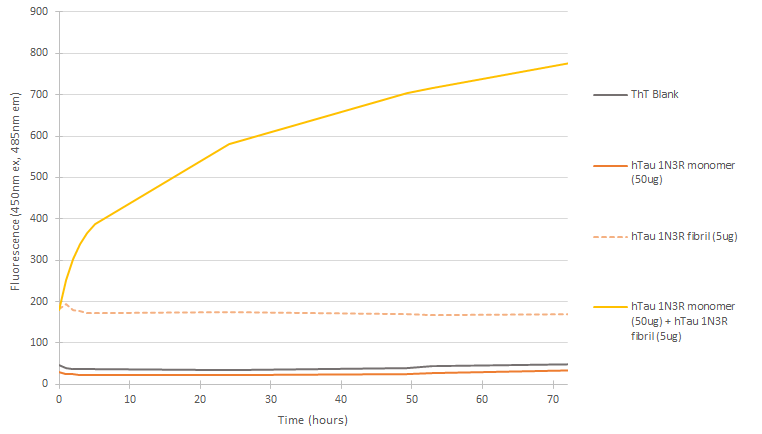
In vitro seeding activity of human tau-381 (1N3R) monomers in ThT assay. Tau 1N3R pre-formed fibrils (SPR-514) seed fibril formation of human tau 1N3R monomers over 72 hours. Reactions (100uL) shaken at 600 rpm in Greiner-Bio 96 Well Non-Binding Cell Culture Microplates, Black (Greiner-Bio Catalog #655900) at 37oC in the presence of 25 uM ThT and 10 uM Heparin and read with an XPS Microplate Reader set at 450nmex/485nmem.





















Reviews
There are no reviews yet.