| Product Name | Tau and Alpha Synuclein Co-Polymer Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
Human Recombinant Tau-352 (fetal 0N3R) and Human Recombinant Alpha Synuclein Co-Polymer Fibrils |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS PAGE, In vitro assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | Total Protein Concentration: 2mg/mL (1mg/ml of tau and 1mg/ml of aSyn) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amino Acid Sequence |
Tau: MAEPRQEFEVMEDHAGTYGLGDRKDQGGYTMHQDQEGDTDAGLKAEEAGIGDTPSLEDEAAGHVTQARMVSKSKDGTGSDDKKAKGADGKTKIATPRGAAPPG QKGQANATRIPAKTPPAPKTPPSSGEPPKSGDRSGYSSPGSPGTPGSRSRTPSLPTPPTREPKKVAVVRTPPKSPSSAKSRLQTAPVPMPDLKNVKSKIGSTENLKHQPGGG KVQIVYKPVDLSKVTSKCGSLGNIHHKPGGGQVEVKSEKLDFKDRVQSKIGSLDNITHVPGGGNKKIETHKLTFRENAKAKTDHGAEIVYKSPVVSGDTSPRHLSNVSSTGSIDMVDSPQLATLADEVSASLAKQGL Asyn: MDVFMKGLSKAKEGVVAAAEKTKQGVAEAAGKTKEGVLYVGSKTKEGVVHGVATVAEKTKEQVTNVGGAVVTGVTAVAQKTVEGAGSIAAATGFVKKDQLGKNEEGAP QEGILEDMPVDPDNEAYEMPSEEGYQDYEPEA |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | Full Length (Tau 0N3R: 1-352 aa, Asyn: 1-140 aa) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | 0N3R: 36.76 kDa, ASYN: 14.46 kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | 1X PBS pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau and Alpha Synuclein Co-Polymer Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-494) |
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding monomers, see catalog# SPR-321 and SPR-490 |
Biological Description
| Alternative Names | MAPT, Tau, Tau40, Fetal Tau, 0N3R, neurofibrillary tangle, paired-helical filament, PHFs, SNCA, NACP, PARK1, asyn, alpha-synuclein, PFFs, mixed fibrils |
| Research Areas | Alzheimer's Disease, Neurodegeneration, Neuroscience, Parkinson's Disease, Synuclein, Tangles & Tau, Multiple System Atrophy |
| Swiss Prot | P10636-2 and P37840-1 |
| Scientific Background |
Tau and alpha-synuclein are two key proteins implicated in neurodegenerative diseases such as Alzheimer’s disease, Parkinson’s disease, and other tauopathies and synucleinopathies. Tau, encoded by the MAPT gene, stabilizes microtubules in neurons, while alpha-synuclein, encoded by SNCA, regulates synaptic vesicle trafficking and neurotransmitter release. Brain-specific tau isoforms vary in the number of N-terminal inserts and C- terminal repeat domains due to alternative splicing of exons; only the shortest isoform of tau, 0N3R, is expressed in the fetal brain during neurogenesis. Under pathological conditions, both proteins misfold and aggregate into fibrillar structures. Recent research has revealed that tau and alpha-synuclein can co-assemble into hybrid fibrils—known as co-polymer fibrils—exhibiting unique structural and biochemical properties distinct from their individual aggregates. These co-polymer fibrils demonstrate enhanced stability, seeding capacity, and neurotoxicity, suggesting a synergistic role in accelerating disease progression. Tau/alpha-synuclein co-polymer fibrils are increasingly used in experimental models to study cross-seeding mechanisms, prion-like propagation, and the convergence of proteinopathies. Their ability to mimic mixed pathologies observed in patients with overlapping clinical features, such as dementia with Lewy bodies and Alzheimer’s disease, makes them a powerful tool for translational research. These hybrid fibrils support the development of targeted therapies aimed at disrupting pathological protein interactions, inhibiting co-aggregation, and enhancing clearance mechanisms. By modeling the complex interplay between tau and alpha-synuclein, co-polymer fibrils provide a high-impact platform for advancing understanding of multifactorial neurodegenerative diseases. |
| References |
1. Goedert et al. Multiple Isoforms of Human Microtubule-associated Protein Tau: Sequences and Localization in Neurofibrilary Tangles of Alzheimer’s Disease. Neuron. 1989;3(4):519-526. 2. Jensen et al. α-synuclein Binds to Tau and Stimulates the Protein Kinase A-catalyzed Tau Phosphorylation of Serine Residues 262 and 356. 1999. JBC. 274(36): 25481-25489. DOI:https://doi.org/10.1074/jbc.274.36.25481 3. Giasson et al. Initiation and Synergistic Fibrillization of Tau and Alpha-Synuclein. Science. 2003; 300: 636-40. DOI: 10.1126/science.1082324 4. Guo et al. Distinct α-synuclein Strains Differentially Promote Tau Inclusions in Neurons. 2013. Cell. 154(1) doi:10.1016/j.cell.2013.05.057. 5. Williams et al. Differential Cross-seeding Properties of Tau and α-synuclein in Mouse Models of Tauopathy and Synucleinopathy. Brain Communications. 2020; 2(2):fcaa090. doi:10.1093/braincomms/fcaa090 6. Pan et al. Tau Accelerates α-synuclein Aggregation and Spreading in Parkinson’s Disease. 2022. Brain. Doi: 10.1093/brain/awac171 |
Product Images
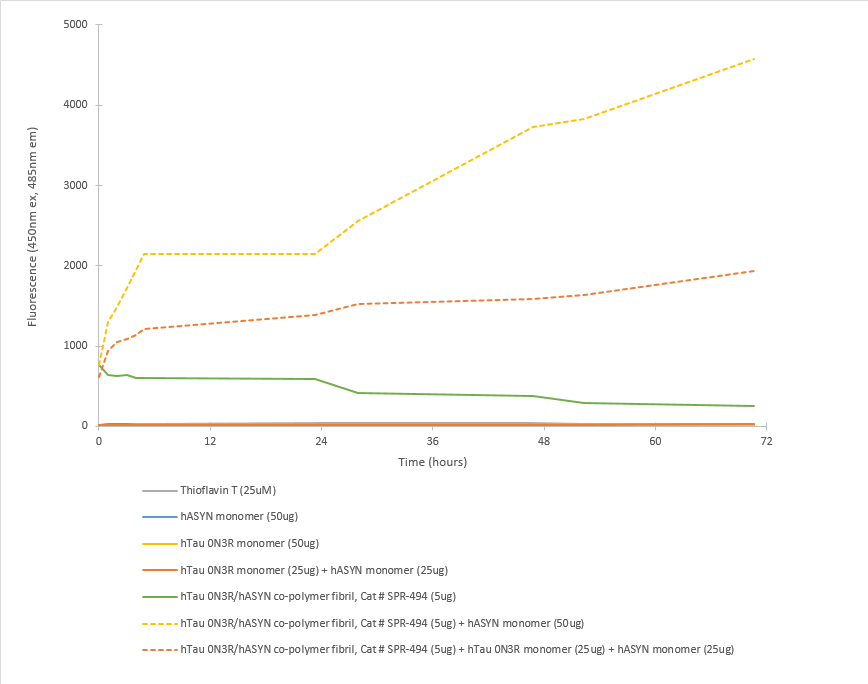
In vitro Seeding activity of Fetal Tau 0N3R & Alpha Synuclein Co-Polymer Fibrils in ThT Assay. Tau 0N3R and Alpha Synuclein co-polymer fibrils seed fibril formation of both Alpha Synuclein monomers and of a mixture of Alpha Synuclein and Tau 0N3R monomers over 72 hours. Reactions (100uL) shaken at 600 rpm in Greiner-Bio 96 Well Non-Binding Cell Culture Microplates, Black (Greiner-Bio Catalog #655900) at 37°C and read with an XPS Microplate Reader set at 450nmex/485nmem.
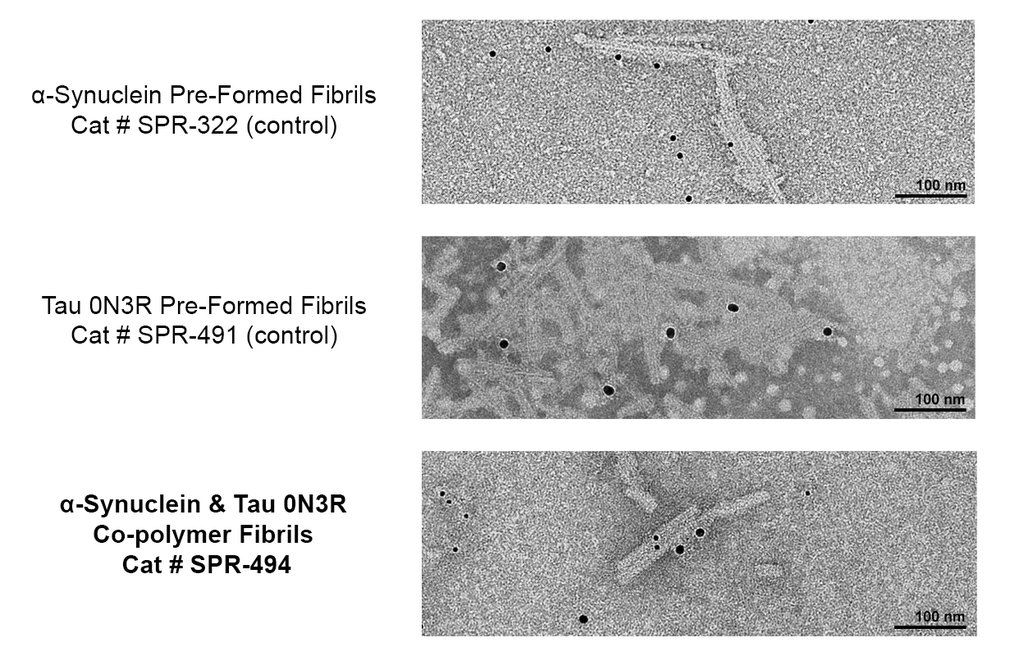
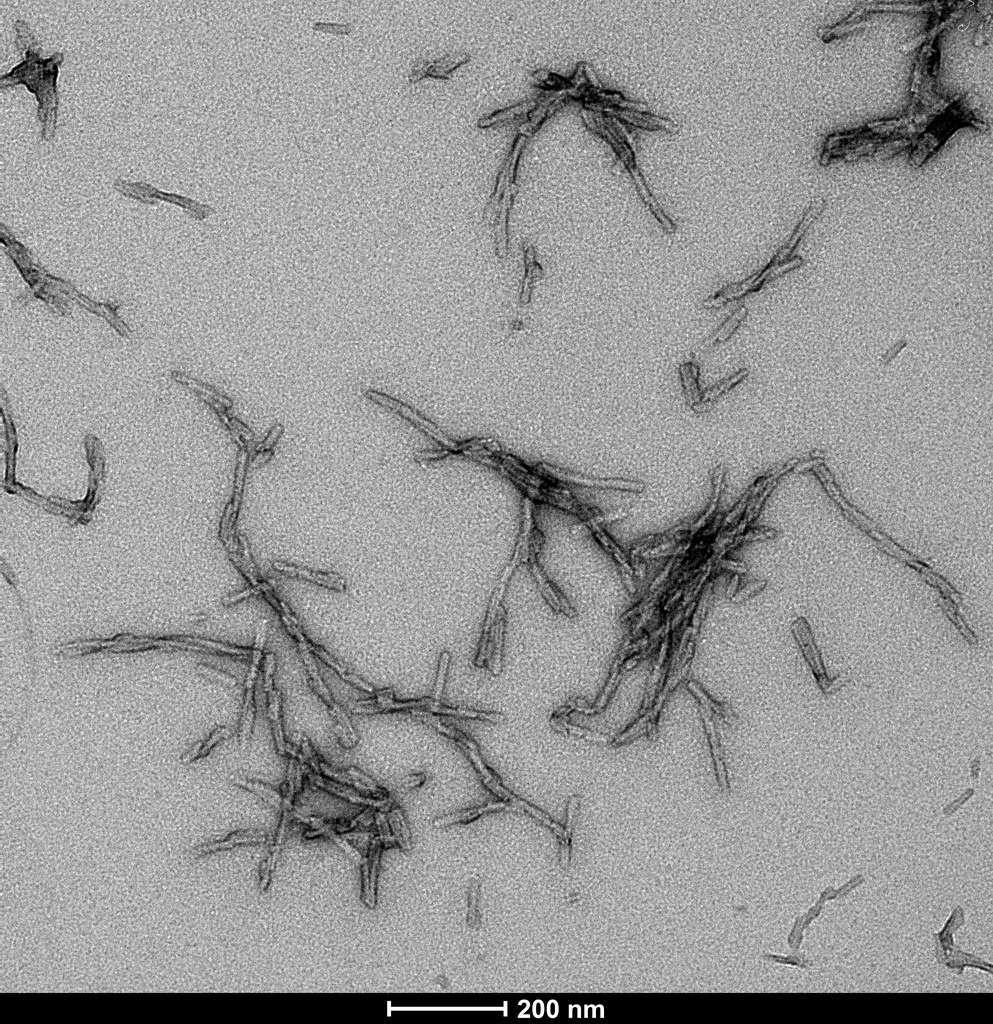
Immuno-TEM of Tau & Alpha Synuclein Co-Polymer Fibrils. Fibrils absorbed onto carbon- coated copper grids. Grids were blocked with 1% BSA and 0.1% Tween-20 in PBS, incubated sequentially with primary antibodies at 20 µg/mL in blocking buffer, washed, then incubated with secondary antibodies at 20 µg/mL in blocking buffer, washed and stained with 2% uranyl acetate as a negative stain. 6nm (asyn) and 12nm (tau) signals only appear together in the same fibril strand for the co-polymer fibril samples, and no antibody cross reactivity is observed in control alpha-synuclein or tau fibrils. Primary antibodies: Mouse anti-human asyn monoclonal (StressMarq SMC-532) and rabbit anti-human tau polyclonal (StressMarq SPC-802); Secondary antibodies: 6nm Colloidal Gold-conjugated Goat Anti-Mouse (Jackson ImmunoResearch 115-195- 146) and 12nm Colloidal Gold-conjugated Goat Anti-Rabbit (Jackson ImmunoResearch 111-205-144).

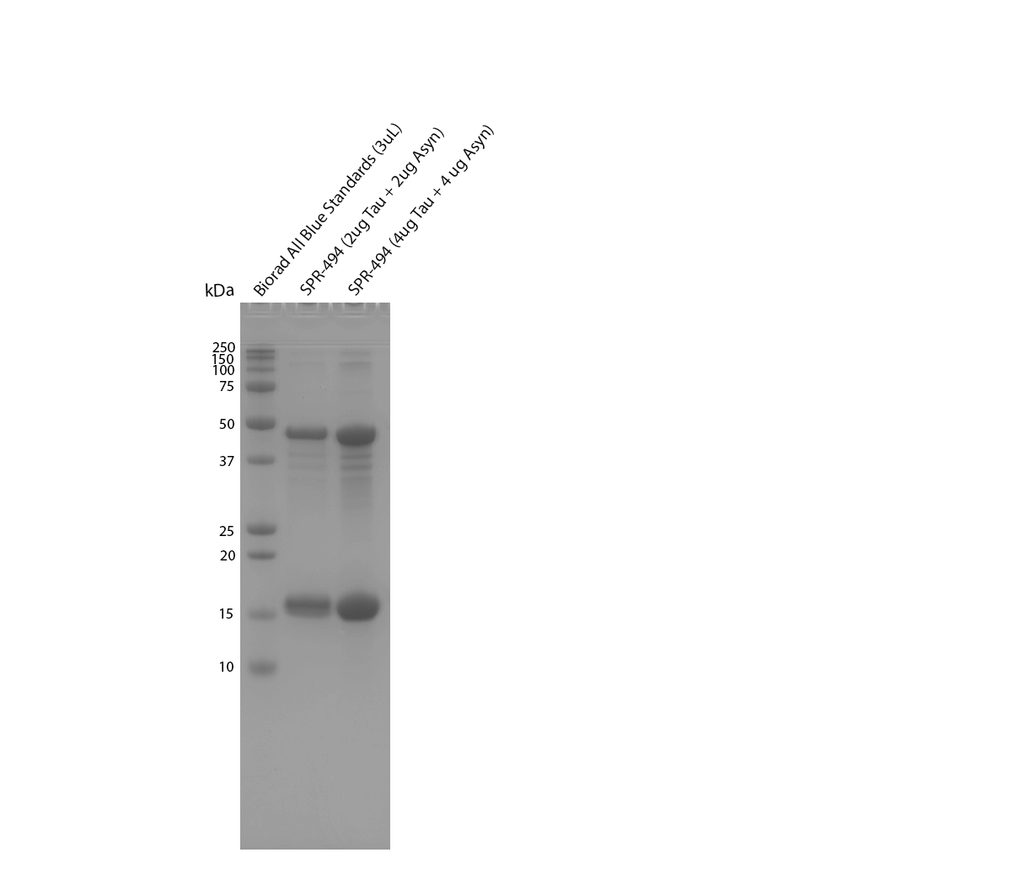




















Reviews
There are no reviews yet.