Properties
| Storage Buffer | 10 mM HEPES, 100 mM NaCl pH 7.4 |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau (K18) P301L Mutant Monomers (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-328) |
| Certificate of Analysis | Certified >95% pure using SDS-PAGE analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For corresponding PFFs, see catalog# SPR-330 |
Biological Description
| Alternative Names | Tau K18 P301L, Tau P301L mutant, microtubule-associated tau, MAPT, MAP, Tau-441, Paired Helical Filament-Tau, Phf-Tau, Neurofibrillary Tangle, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1, Isoform 4, tubulin-associated unit |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Gene ID | 4137 |
| Swiss Prot | P10636 |
| Scientific Background |
Tau is a microtubule-associated protein encoded by the MAPT gene, essential for stabilizing neuronal microtubules and maintaining axonal transport. The K18 fragment of tau, comprising the four microtubule-binding repeat domains, is a widely used model for studying tau aggregation. Within this domain, the P301L mutation—where proline is substituted by leucine at position 301—is strongly associated with familial frontotemporal dementia and other tauopathies. Tau (K18) P301L mutant monomers exhibit altered conformational dynamics that reduce microtubule binding affinity and increase aggregation propensity. These structural changes promote the formation of toxic oligomers and fibrils, which disrupt cytoskeletal integrity, impair axonal transport, and trigger neuroinflammatory responses. Even in their monomeric state, P301L tau variants display enhanced seeding activity, initiating pathological cascades that precede overt neurofibrillary tangle formation. Experimental models using P301L mutant tau monomers reveal early synaptic dysfunction, mitochondrial stress, and progressive neuronal loss. These monomers also influence tau phosphorylation patterns and proteolytic processing, contributing to the accumulation of neurotoxic species. Their ability to self-assemble and propagate misfolded tau across neural networks underscores their role as primary instigators of tau-mediated neurodegeneration. Targeting the early misfolding and aggregation of P301L tau monomers offers a promising therapeutic strategy. Interventions aimed at stabilizing native tau structure, inhibiting seed formation, or enhancing clearance pathways are under active investigation to halt disease progression in tauopathies. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatr. Psych.-Gerichtl. Med. 64, 146–148 (1907) 3. Matsumoto, G. et al. (2018). Int J Mol Sci. 19, 1497. 4. Goedert, M. and Spillantini, M. G. (2017). Mol Brain. 10:18. 5. Guo, J. and Lee, M.Y. (2013). FEBS Lett. 587(6): 717-723. 6. Alberici, A. et al. (2004). J Neurol Neurosurg Psychiatry. 75:1607-1610. |
Product Images
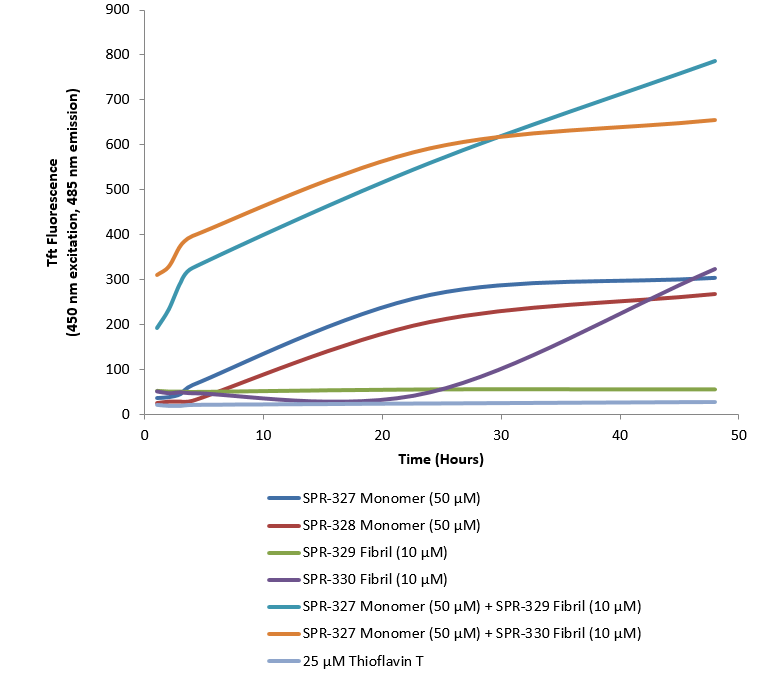
Thioflavin T is a fluorescent dye that binds to beta sheet-rich structures such as those in tau fibrils. Upon binding, the emission spectrum of the dye experiences a red-shift, and increased fluorescence intensity. Thioflavin T emission curves show increased fluorescence (correlated to tau aggregation) in tau K18 P301L monomers (SPR-328) over time. Thioflavin T ex = 450 nm, em = 485 nm. 10 uM heparin was added to each well.






















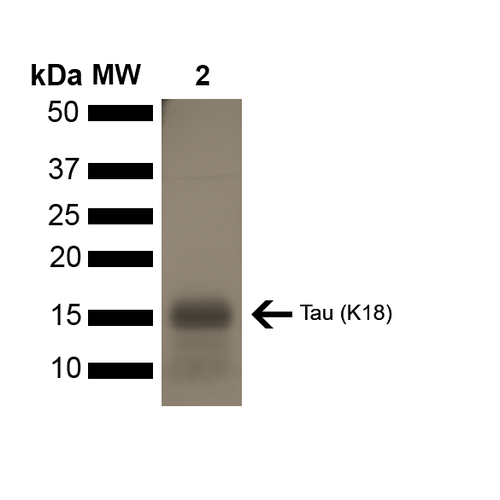
Reviews
There are no reviews yet.