| Product Name | Tau-352 (fetal 0N3R) Wild-Type Pre-formed Fibrils | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description |
Human Recombinant Tau-352 (fetal 0N3R) Wild-Type PFFs |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | WB, SDS-PAGE, In vitro Assay | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration | 2 mg/ml or 5 mg/ml | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Conjugates |
No tag
StreptavidinProperties:
Biotin
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature | Recombinant | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Species | Human | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expression System | E. coli | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amino Acid Sequence | MAEPRQEFEVMEDHAGTYGLGDRKDQGGYTMHQDQEGDTDAGLKAEEAGIGDTPSLEDEAAGHVTQARMVSKSKDGTGSDDKKAKGADGKTKIATPRGAAPPGQKGQANATRIPAKTPPAPKTPPSSGEPPKSGDRSGYSSPGSPGTPGSRSRTPSLPTPPTREPKKVAVVRTPPKSPSSAKSRLQTAPVPMPDLKNVKSKIGSTENLKHQPGGGKVQIVYKPVDLSKVTSKCGSLGNIHHKPGGGQVEVKSEKLDFKDRVQSKIGSLDNITHVPGGGNKKIETHKLTFRENAKAKTDHGAEIVYKSPVVSGDTSPRHLSNVSSTGSIDMVDSPQLATLADEVSASLAKQGL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
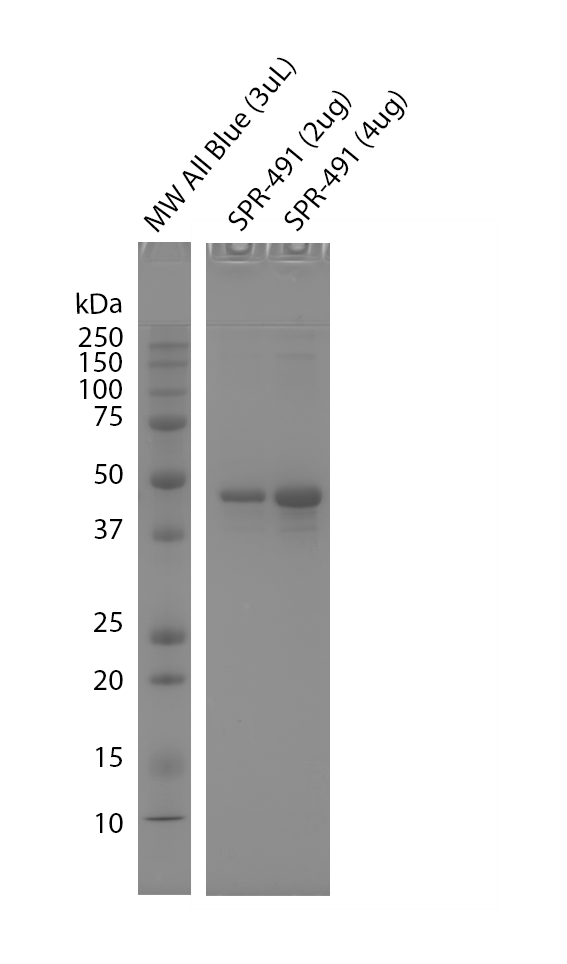
| Purity | >95% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Resources | Sonication Protocol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Length | Full Length (1-352 aa) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Protein Size | 37 kDa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Properties
| Storage Buffer | 10 mM Hepes pH 7.4, 100 mM NaCl |
| Storage Temperature | -80ºC |
| Shipping Temperature | Dry Ice. Shipping note: Product will be shipped separately from other products purchased in the same order. |
| Purification | Ion-exchange Purified |
| Cite This Product | Human Recombinant Tau-352 (fetal 0N3R) Wild-Type Pre-formed Fibrils (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SPR-491) |
| Certificate of Analysis | Protein certified >95% pure on SDS-PAGE & Nanodrop analysis. Low endotoxin <5 EU/mL @ 2mg/mL. |
| Other Relevant Information | For best results, sonicate immediately prior to use. Refer to the Neurodegenerative Protein Handling Instructions on our website, or the product datasheet for further information. Monomer source is catalog# SPR-490. |
Biological Description
| Alternative Names | Tau, Tau aggregate, microtubule-associated Tau, MAPT, MAP, microtubule-associated, Truncated Tau, Paired Helical Filament-Tau, Phf-Tau, Neurofibrillary Tangle, G Protein Beta1/Gamma2 Subunit-Interacting Factor 1, Isoform 2, tubulin-associated unit, 95-amino acid Tau fragment, Tau-352, 0N3R Tau, fetal Tau, Tau PFFs |
| Research Areas | Alzheimer's Disease, Axon Markers, Cell Markers, Cell Signaling, Cytoskeleton, Microtubules, MT Associated Proteins, Neurodegeneration, Neuron Markers, Neuroscience, Tangles & Tau |
| Cellular Localization | Axolemma, Axolemma Plasma Membrane, Axon, Cell Body, Cell membrane, Cytoplasm, Cytoplasmic Ribonucleoprotein Granule, Cytoplasmic Side, Cytoskeleton, Cytosol, Dendrite, Growth cone, Microtubule, Microtubule Associated Complex, Neurofibrillary Tangle, Neuronal Cell Body, Nuclear Periphery, Nuclear Speck, Nucleus, Peripheral membrane protein, Plasma Membrane, Tubulin Complex |
| Accession Number | NP_058525.1 |
| Gene ID | 4137 |
| Swiss Prot | P10636-2 |
| Scientific Background |
Tau protein, encoded by the MAPT gene (UniProt ID: P10636), is essential for microtubule stabilization and neuronal function. The Tau-352 isoform, also known as 0N3R, is a fetal variant composed of three microtubule-binding repeats and no N-terminal inserts. This isoform is predominantly expressed during early brain development and is replaced by longer adult isoforms postnatally. Pre-formed fibrils (PFFs) generated from wild-type Tau-352 replicate early aggregation events and provide a unique model for studying the structural and functional properties of fetal Tau in the context of neurodegenerative disease. Although less aggregation-prone than adult isoforms, 0N3R Tau can form β-sheet-rich fibrils under pathological conditions, contributing to tauopathy progression. Tau-352 PFFs are used in experimental systems to investigate the mechanisms of Tau seeding, propagation, and neurotoxicity. Their relevance extends to disorders such as Alzheimer’s disease and Pick’s disease, where altered isoform ratios and early aggregation events are implicated in disease onset. These fibrils enable high-resolution studies of Tau’s prion-like behavior and support the development of therapeutic strategies aimed at preventing aggregation and promoting clearance. By modeling the transition from developmental Tau to pathogenic aggregates, Tau-352 wild-type PFFs offer a valuable platform for understanding early-stage tauopathies and advancing neurodegenerative disease research. |
| References |
1. www.alz.org/alzheimers-dementia/facts-figures 2. Goedert et al. Multiple isoforms of human microtubule-associated protein tau: Sequences and localization in neurofibrilary tangles of Alzheimer’s disease. Neuron. 1989;3(4):519-526. 3. Shahpasand-Kroner et al. Three-repeat and four-repeat tau isoforms for different oligomers. Prot. Sci. 2021;doi: 10.1002/pro4257 4. Dregni, et al. Inclusion of the C‑Terminal Domain in the β‑Sheet Core of Heparin-Fibrillized Three-Repeat Tau Protein Revealed by Solid-State Nuclear Magnetic Resonance Spectroscopy. JACS. 2021. https://doi.org/10.1021/jacs.1c03314 |
Product Images
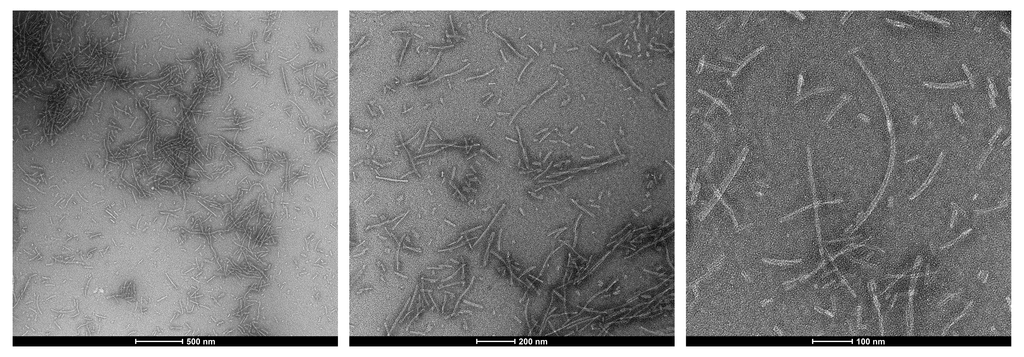
TEM of Fetal Tau 0N3R fibrils. Negative stain transmission electron microscopy images acquired at 80 Kv on carbon coated 400 mesh copper grids using phosphotungstic acid and uranyl acetate stain. Scale bar = 500, 200 and 100 nm (left to right). Method: Samples were prepared for examination in the transmission electron microscope using the ‘direct application method’ (Doane and Anderson 1987).
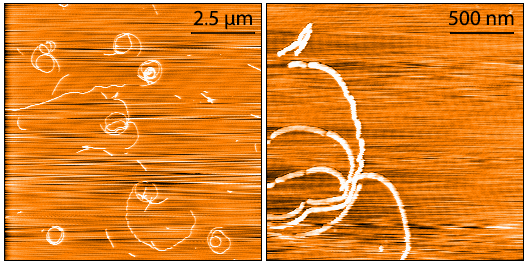
AFM of Fetal Tau 0N3R fibrils. Atomic force microscopy analysis of 1.0 mg/mL samples diluted to 0.1 mg/mL in dH2O, mounted on freshly cleaved mica, washed, dried and analyzed with tapping mode. Representative images are 10 x 10 µm x-y (left) and 2 x 2 µm x-y (right) both with a z-range of 6 nm. Note: AFM images display significant twisting and curvature not observed under TEM.
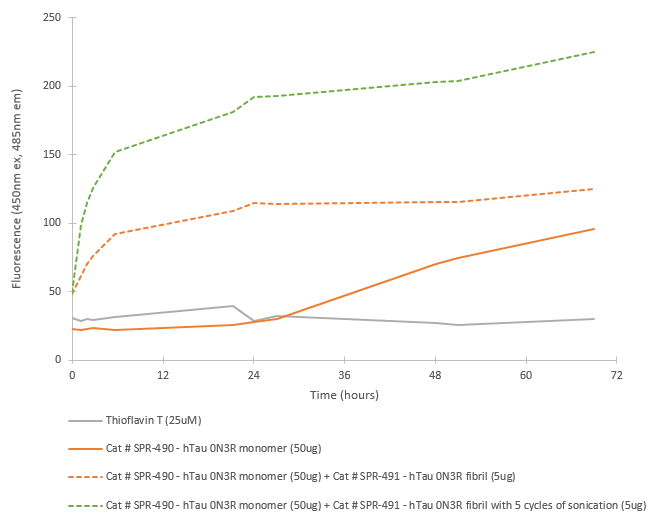
Fibril formation activity of Fetal Tau 0N3R monomers seeded by pre-formed fibrils in ThT seeding assay. Tau 0N3R pre-formed fibrils seed fibril formation in Tau 0N3R monomers. The graph shows an increased ThT signal in fibrils with 5 cycles of sonication prior to seeding. 10uM heparin and 1mM DTT was added to each well.





















StressMarq Biosciences :
Based on validation through cited publications.