| Product Name | T0901317 |
| Description |
Amyloid Beta Regulator |
| Purity | >98% (TLC); NMR (Conforms) |
| CAS No. | 293754-55-9 |
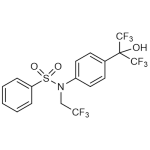
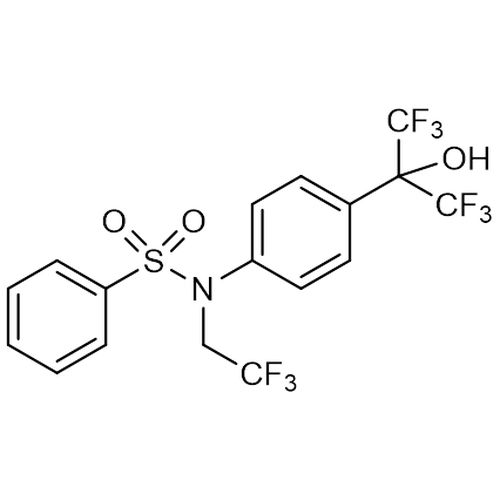
| Molecular Formula | C17H12F9NO3S |
| Molecular Weight | 481.3 |
| Field of Use | Not for use in humans. Not for use in diagnostics or therapeutics. For in vitro research use only. |
Properties
| Storage Temperature | -20ºC |
| Shipping Temperature | Shipped Ambient |
| Product Type | Regulator |
| Solubility | May be dissolved in DMSO (50 mg/ml); or ethanol (50 mg/ml) |
| Source | Synthetic |
| Appearance | Beige to off-white powder |
| SMILES | OC(C1=CC=C(C=C1)N(CC(F)(F)F)S(=O)(=O)C1=CC=CC=C1)(C(F)(F)F)C(F)(F)F |
| InChI | InChI=1S/C17H12F9NO3S/c18-14(19,20)10-27(31(29,30)13-4-2-1-3-5-13)12-8-6-11(7-9-12)15(28,16(21,22)23)17(24,25)26/h1-9,28H,10H2 |
| InChIKey | SGIWFELWJPNFDH-UHFFFAOYSA-N |
| Safety Phrases | Not a hazardous substance or mixture. |
| Cite This Product | T0901317 (StressMarq Biosciences | Victoria, BC CANADA | Catalog# SIH-643) |
Biological Description
| Alternative Names | N-(2,2,2-Trifluoroethyl)-N-[4-[2,2,2-trifluoro-1-hydroxy-1-(trifluoromethyl)ethyl]phenyl]-benzenesulfonamide |
| Research Areas | Alzheimer's Disease, Apoptosis, Cancer, Neurodegeneration, Neuroscience |
| PubChem ID | 447912 |
| Scientific Background |
T0901317 is a potent, high-affinity agonist of liver X receptors LXRα and LXRβ (EC₅₀ ≈ 50 nM). Of particular relevance to neuroscience, T0901317 has been shown to decrease amyloid‑β production in a mouse model of Alzheimer’s disease, highlighting its potential utility in neurodegenerative disease research. This effect is thought to be mediated, at least in part, through upregulation of ABCA1, a key reverse cholesterol transporter implicated in brain lipid homeostasis and amyloid processing. Beyond its central nervous system effects, T0901317 inhibits intestinal cholesterol absorption via ABCA1 induction and acts as an inverse agonist at CAR2. The compound has also been reported to inhibit cellular senescence in endothelial cells. Notably, T0901317 displays off‑target activity as an FXR agonist, albeit at lower potency than LXR, necessitating the use of appropriate experimental controls when employing this compound as a pharmacological tool. |
| References |
1. Repa JJ et al. Science. 2000;289:1524 2. Kanno Y et al. J. Toxicol. Sci. 2013;38:309 3. Houck KA et al. Mol. Genet. Metab. 2004;83:184 4. Koldamova RP et al. J. Biol. Chem. 2005;280:4079 5. Hayashi T et al. Proc. Natl. Acad. Sci. USA. 2014;111:1168 |

Reviews
There are no reviews yet.